Ozone Therapy: A Promising Alternative Treatment with Potential Benefits
MONDAY, 24 APRIL 2023 / PUBLISHED IN BLOG
Ozone therapy is a promising alternative medical treatment that has gained popularity in recent years. It involves the administration of ozone, a form of oxygen, to treat various medical conditions, ranging from chronic pain to infections. Although the scientific evidence on the effectiveness of ozone therapy remains limited, many people claim to have experienced significant health benefits from this alternative treatment. In this article, we’ll explore the potential benefits and risks of ozone therapy, as well as the latest research on this alternative medical treatment.
What is Ozone Therapy?
Ozone therapy is a safe, non-invasive, and effective alternative medical treatment that has been used for over a century. It involves the administration of ozone, a gas made up of three oxygen atoms, to the body in various forms, such as injections, insufflations, or topical applications. Ozone is a powerful oxidant that can damage cells and tissues, which can have positive or negative effects, depending on the form and amount of ozone used.
One of the key benefits of ozone therapy is its ability to improve blood circulation and oxygen delivery to the tissues, which can help reduce inflammation, support wound healing, and boost immune function. Additionally, ozone therapy is believed to have antimicrobial and antiviral properties, which may help fight infections caused by bacteria, viruses, and fungi.
Potential Benefits of Ozone Therapy
Ozone therapy has been used to treat a wide range of medical conditions, including:
- Chronic Pain: Ozone therapy may help reduce pain and inflammation in conditions such as osteoarthritis, herniated discs, and fibromyalgia.
- Infections: Ozone therapy may help fight infections caused by bacteria, viruses, and fungi, including Lyme disease, hepatitis, and HIV.
- Autoimmune Diseases: Ozone therapy may help reduce inflammation and improve immune function in conditions such as multiple sclerosis, rheumatoid arthritis, and lupus.
- Skin Conditions: Ozone therapy may help treat various skin conditions, including eczema, psoriasis, and acne, by improving blood circulation and oxygenation.
Moreover, ozone therapy is a safe and non-invasive treatment that doesn’t have the side effects commonly associated with traditional medical treatments. It’s also relatively inexpensive and can be administered in a variety of settings, including clinics, hospitals, and private practices.
Latest Research on Ozone Therapy
Although the scientific evidence on the effectiveness and safety of ozone therapy remains limited, some recent studies have suggested that ozone therapy may have potential health benefits in certain conditions.
- Immunomodulatory Properties: A study published in the Cuban journal of immunology describes the immunomodulatory properties of ozone therapy by characterizing the biological effects of ozone on immune system cells, soluble mediators, and other cell types.
- Pain Management: A 2022 review on ozone therapy in pain medicine highlights the high safety of pain treatments with ozone therapy, especially modern medical ozone generators with great precision.
- Diabetic Pain Relief: A 2020 study on systemic ozone therapy in insulin-dependent diabetic patients found that the analgesic and anti-inflammatory properties of ozone make it one of the most efficient therapeutic tools for controlling chronic pain in insulin-dependent diabetic patients.
Conclusion
Ozone therapy is a promising alternative medical treatment that has been used to treat various medical conditions. Many people claim to have experienced significant health benefits from this alternative treatment. Furthermore, ozone therapy is a safe and non-invasive treatment that doesn’t have the side effects commonly associated with traditional medical treatments.
If you’re interested in ozone therapy, make sure to do your research, ask questions, and consider this alternative medical treatment as a potential option for your health concerns. It’s important to stay informed about the latest developments in stem cell therapy. You can learn more about regenerative medicine and stem cells by enrolling in our international certification program at www.issca.us.
- Published in Blog
Personalized Regenerative Medicine: The Future of Regenerative Medicine
MONDAY, 17 APRIL 2023 / PUBLISHED IN BLOG
Regenerative medicine is a rapidly growing field that focuses on repairing or replacing damaged or diseased tissues and organs using stem cells, growth factors, and other advanced therapies. The effectiveness of this approach has been demonstrated in treating a wide range of conditions, including heart disease, diabetes, joint pain, and neurological disorders. One of the major challenges of regenerative medicine, however, is that not every patient responds to the same treatment. This is where personalized regenerative medicine comes into play.
What is Personalized Regenerative Medicine?
Personalized regenerative medicine involves tailoring treatments to meet the unique needs of each patient based on their genetic profile, lifestyle, and medical history. This approach recognizes that no two patients are the same and that a one-size-fits-all approach to regenerative medicine is not always effective.
As part of personalized regenerative medicine, advanced technologies such as genomics, proteomics, and metabolomics are used to analyze a patient’s unique biological profile. Regenerative medicine centers utilize this information to develop personalized treatment plans tailored to their individual needs and conditions, ranging from stem cell therapy to gene therapy.
Why is Personalized Regenerative Medicine the Future of Regenerative Medicine?
There are several reasons why personalized regenerative medicine is the future of regenerative medicine:
More Effective Treatments
By tailoring treatments to individual patients, personalized regenerative medicine has the potential to be much more effective than traditional one-size-fits-all approaches.
Improved Patient Outcomes
Because personalized regenerative medicine takes into account the unique needs and conditions of each patient, it has the potential to improve patient outcomes and reduce the risk of complications.
Lower Healthcare Costs
By providing more effective treatments and reducing the risk of complications, personalized regenerative medicine has the potential to lower healthcare costs in the long run.
Better Understanding of Disease
Personalized regenerative medicine involves analyzing a patient’s unique biological profile, which can help researchers and healthcare providers better understand the underlying mechanisms of disease.
Advancements in Technology
As technology continues to advance, personalized regenerative medicine will become even more precise and effective, leading to even better patient outcomes.
Conclusion
The concept of personalized regenerative medicine is an exciting new approach to regenerative medicine that has the potential to revolutionize the healthcare industry. Individualized regenerative medicine has the potential to be more effective, improve patient outcomes, lower healthcare costs, and provide better understanding of disease by tailoring treatments to the individual patient. As technology continues to advance, we can expect to see even more exciting developments in this field in the years to come.
You can learn more about regenerative medicine and stem cells by enrolling in our international certification program at www.issca.us.
- Published in Blog
Revolutionizing Neurodegenerative Care: The Advancements in Stem Cell Therapy
MONDAY, 13 MARCH 2023 / PUBLISHED IN BLOG
Neurodegenerative diseases, including Alzheimer’s disease, Parkinson’s disease, Huntington’s disease, and amyotrophic lateral sclerosis, are a group of incurable neurological disorders characterized by the chronic progressive loss of different neuronal subtypes. The conditions are debilitating and affect millions of people worldwide. Despite its increasing prevalence among the ever-increasing aging population, little progress has been made in the coincident immense efforts towards development of therapeutic agents. However, recent advancements in stem cell therapy, including stem cell-derived exosomes, neurotrophic factors (NTFs), and their combination, show promise as potential therapeutic agents in neurodegenerative diseases.
The Promise of Stem Cell Therapy
In recent years, stem cells have emerged as one of the most active research therapeutic tools for many diseases. Stem cells are undifferentiated cells that have the ability to develop into pluripotent stem cells and adult stem cells. All stem cells have the potential for continuous self-renewal, high proliferation, and multidirectional differentiation into various cell types to replace degenerated or dead cells. In the context of neurodegenerative diseases, stem cells can be used to replace lost or damaged neurons, provide neuroprotection, and promote neural repair. These properties make stem cells an attractive option for treating neurodegenerative diseases, as they have the potential to slow or even reverse the progression of these conditions.
Advantages of Stem Cell Therapy
One of the key benefits of stem cell therapy for neurodegenerative diseases is that it offers a non-invasive, minimally-invasive, and non-toxic alternative to traditional treatments. Unlike drugs and surgery, stem cell therapy does not produce adverse side effects and can be administered in a safe and controlled manner. In clinical trials, stem cell therapy has been shown to be effective in reducing symptoms and improving quality of life for patients with neurodegenerative diseases.
Alzheimer’s Disease (AD)
Alzheimer’s disease (AD) is a progressive neurodegenerative disease and the most common form of dementia, affecting approximately 55 million people worldwide. Cognitive impairment is a hallmark of AD. Currently, drug therapies can only delay symptoms, but not relieve disease pathology or progression. Researchers have demonstrated that neurons derived from stem cells can integrate with existing neural networks and repair damaged neurons within the host brain, improving learning and memory deficits. NTFs can improve AD symptoms and provide neuroprotective effects.
Parkinson’s Disease (PD)
The second most common neurodegenerative disorder is Parkinson’s disease (PD). The motor symptoms of PD mainly include rest tremor, rigidity, bradykinesia, and postural instability, while common nonmotor symptoms include neuropsychiatric and sleep disorders as well as sensory and autonomic dysfunction. At present, there is no cure for PD or disease-modifying therapy. It is common for symptom-relief medications to only provide partial relief and elicit side effects such as motor complications, gastrointestinal problems, and neurological issues. In spite of the fact that these treatments do not address the underlying pathology, alternative therapies, especially those based on stem cells and NTFs, are still being pursued intensively.
Huntington’s Disease (HD)
Huntington’s disease (HD) is characterized by motor, cognitive, and psychiatric dysfunctions. The study of multiple possible neurodegenerative mechanisms of HD is currently underway, and this knowledge is expected to contribute to the development of new HD treatments. The abilities of stem cells to rescue or replace the damaged and dying neurons, and to prevent further cell damage and death, make stem cell-based therapies promising for treatment of this neurodegenerative disease.
Amyotrophic Lateral Sclerosis (ALS)
As a neurodegenerative disorder, amyotrophic lateral sclerosis (ALS) involves progressive degeneration of both upper and lower motor
neurons, resulting in palsy and death within 3-5 years of onset. Currently, only two disease-modifying medicines are available, each showing benefit to a limited number of patients. Stem cell-based therapy holds great promise for treating ALS by providing both cell replacement and NTF delivery to target the multiple pathologies. Stem cells available for ALS treatment include NSCs, MSCs, embryonic stem cells, induced pluripotent stem cells, and olfactory ensheathing stem cells.
The Future of Stem Cell Therapy in Neurodegenerative Diseases
While stem cell therapy is still in the early stages of development, it holds enormous promise as a future treatment for neurodegenerative diseases. A number of studies have demonstrated that stem cells and NTFs offer considerable therapeutic potential, especially when they are used together. In addition to replenishing target neurons, stem cells combined with NTFs can produce neurotrophins to improve the microenvironment that promotes nerve repair and regeneration. As research continues, we can expect to see even more exciting advancements in this field, bringing hope to those affected by these debilitating conditions.
Conclusion
Stem cell therapy represents a revolutionary approach to treating neurodegenerative diseases, offering a non-toxic, minimally-invasive alternative to traditional treatments. With ongoing research and development, the potential to improve patient outcomes and quality of life is immense. It’s important to stay informed about the latest developments in stem cell therapy. You can learn more about regenerative medicine and stem cells by enrolling in our international certification program at www.issca.us.
- Published in Blog
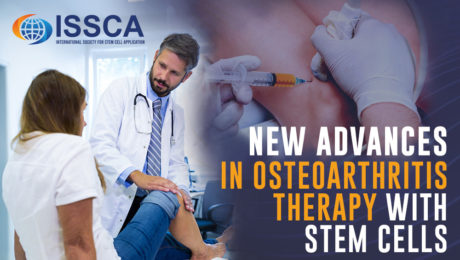
New advances in osteoarthritis therapy with stem cells
FRIDAY, 18 NOVEMBER 2022 / PUBLISHED IN BLOG
Osteoarthritis is a rheumatic pathology that damages the articular cartilage. By joining two bones through the joint capsule, the joints are able to move, providing us with functional autonomy. An inner fluid called synovial fluid is usually found within joints, which is produced by the synovial membrane. Articular cartilage covers the ends of the bones that form the joint. As a result of damage to this articular cartilage, pain, stiffness, and functional impairment occur. Osteoarthritis is the most common joint disorder, usually beginning between the ages of 40 and 50, affecting to some degree almost everyone over the age of 80. Typically, osteoarthritis affects the spine, shoulders, fingers, hips, knees, and toe joints.
Potential of Stem Cell Therapies
Stem cell therapies have the potential to treat a broad spectrum of diseases, including osteoarthritis, rhizarthrosis, diabetes, neurodegenerative diseases, spinal cord injuries, and heart disease. By utilizing stem cells, regenerative medicine is capable of repairing tissues in affected areas. The main difference between lipogem therapy and other treatments for osteoarthritis is that lipogem therapy regenerates cartilage, avoids surgery and its sequelae, and improves the quality of life for patients.
Advances in Sports Medicine and Traumatology
The potential for medical treatments with stem cells and their by-products is currently very high. In the field of sports medicine and traumatology, one of the most outstanding advances has been made for the first time in decades recently: Spanish scientists have achieved a degree of tendon regeneration in 100% of injured patients, resulting in a decrease in pain and a return to sport within two months, and just six months after the trial was completed.
A research performed by the Institute of Regenerative Tissue Therapy (ITRT), published by the prestigious American Journal of Sports Medicine, demonstrates how this therapy regenerates chronic lesions in the patellar tendon and opens up a new therapeutic option for this tissue, which was considered impossible to regenerate.
Adipose Tissue-Derived MSCs
In most patients, fat tissue can be harvested minimally invasively (under local or general anesthesia), providing a highly viable MSC population regardless of donor age. Similar to MSCs derived from other tissues, adipose tissue-derived MSCs have regenerative potential. As osteoarthritis is a very common joint disease, and knee osteoarthritis is the most common form, it is necessary to review scientific literature on osteoarthritis treatments with stem cells, like lipogems.
Lipogems Therapy
Lipogems therapy is a novel procedure that enhances the body’s natural ability to heal itself through the innovative power of science and biotechnology. The Lipogems method involves injecting mesenchymal stem cells into the joints. Adipose-derived mesenchymal stem cells have enormous regenerative potential. They also have a regenerative capacity independent of their age. Even older individuals can benefit from this procedure.
Injection of mesenchymal stem cells into the knee, particularly in the early stages of osteoarthritis, can stop the process of inflammation and degeneration, especially in the less advanced stages of the disease. In addition to preventing progressive physical deterioration of the articular cartilage, this treatment contributes significantly to a patient’s well-being and prevents the installation of knee prostheses.
Patellar Tendinopathy: Physiotherapeutic Treatment and Stem Cell Therapy
Injuries to the patellar tendon that connects the kneecap to the tibia are known as patellar tendinopathy or patellar tendinitis. The patellar tendon works with the muscles in the front of the thigh to extend the knee so you can kick, run, and jump. Athletes who perform frequent jumping in their sports, such as basketball and volleyball, are most likely to suffer from patellar tendonitis. However, people who don’t engage in jumping sports may develop patellar tendonitis. Patients with patellar tendinitis usually begin treatment with physical therapy to stretch and strengthen their knee muscles.
Strength Training and Stem Cell Therapy for Tendinopathies
Strength training with eccentric resistance is one of the most common treatments for tendinopathies. Alternatively, it has been demonstrated that bone marrow-derived mesenchymal stem cells (MSCs) can regenerate injured patellar tendons. Within six months of treatment, it has been observed that the structure of this tissue – which is always difficult to treat – is restored, reaching a regeneration of 40% in all injured persons, with a gradual improvement that eventually becomes complete.
Combination Therapies for Patellar Tendinopathy
It has been found that traditional management methods, including isometric or eccentric exercises, shock wave therapy, and even surgery, are not effective. As part of a rehabilitation program in chronic patellar tendinopathy, autologous expanded bone marrow mesenchymal stem cells (BM-MSC) or leukocyte-poor platelet-rich plasma (Lp-PRP) may be effective in reducing pain and improving activity levels. Traditional management, which includes isometric or eccentric exercises, shock wave therapy, and even surgery, has limited success. A combination of autologous expanded bone marrow mesenchymal stem cells (BM-MSCs) and leukocyte-poor platelet-rich plasma (Lp-PRP) and rehabilitation may reduce pain and improve activity levels in active participants with chronic patellar tendinopathy.
To learn more about stem cells, cellular therapies, and keep up to date with all the information about regenerative medicine and its advances, sign up for our international certification in regenerative medicine at www.issca.us.
- Published in Blog
Regenerative Medicine – An Overview On Stem Cell Therapy
THURSDAY, 10 NOVEMBER 2022 / PUBLISHED IN BLOG
Stem cell therapy is a form of regenerative medicine designed to repair damaged cells within the body by reducing inflammation and modulating the immune system. This phenomenon makes stem cell therapy a viable treatment option for a variety of medical conditions.
What is Stem Cell Therapy?
The term stem cell therapy refers to any treatment involving the use of viable human stem cells, including embryonic stem cells (ESCs), induced pluripotent stem cells (iPSCs), and adult stem cells. Stem cells have the unique ability to differentiate into specific cell types necessary for repairing diseased tissues, making them ideal for tissue and organ transplantation.
The Hierarchy of Stem Cells
In general, stem cells fall into three categories based on their differentiation potential:
- Totipotent Cells: The most basic and capable of forming a complete embryo and extraembryonic tissues.
- Pluripotent Cells (Embryonic Stem Cells): Can differentiate into any of the three embryonic germ layers (ectoderm, mesoderm, and endoderm).
- Adult Stem Cells: Found in specialized tissues throughout the body, capable of differentiating into specific cell types related to their tissue of origin.
Adult stem cells are considered the gold standard for stem cell-based therapies due to their versatility and ability to maintain homeostasis throughout an organism’s lifespan.
Stem Cell Research for Treating Disease
Stem cell research has made significant advancements, particularly with induced pluripotent stem cells (iPSCs), which are generated from adult cells and share properties with embryonic stem cells. This innovation has enhanced disease identification and treatment possibilities, particularly in autologous transplants using patient-matched cells.
Applications of Stem Cells
Stem cells hold potential for treating diseases such as diabetes and heart disease due to their regenerative capabilities. Ongoing research aims to better understand how stem cells can be utilized in regenerative and reparative medicine to effectively treat various conditions.
Future Directions in Stem Cell Research
Stem cell studies continue to expand our knowledge of cell development and regeneration. Researchers are leveraging stem cells to test new drugs, develop model systems for studying normal development, and investigate the causes of birth defects.
To learn more about stem cells, cellular therapies, and new medical protocols using exosomes, sign up for our international certification in regenerative medicine at www.issca.us.
- Published in Blog
ISSCA Members to Present in the XXV Congreso Internacional de Medicina, Cirugia Estética y Obesidad
The XXV Congreso Internacional de Medicina, Cirugia Estética y Obesidad will take place in CDMX, México on July 13,14 and 15, 2022. The International Society for Stem Cell Application (ISSCA) will be actively taking part in the congress. The ISSCA is a multidisciplinary community of physicians and scientists with a mission to advance the science, technology, and practice of regenerative medicine to treat diseases and lessen human suffering through science, technology, and regenerative medicine. Several ISSCA members will be presenting or giving demonstrations on the latest protocols and technologies in regenerative medicine.
- Benito Novas, the managing director of the ISSCA from the USA, will give a talk about Digital Marketing in Cash-Based Practices. Benito Novas is a global entrepreneur, manager, and keynote speaker, who specializes in marketing focused on biotechnology, life sciences, and healthcare development. He has served as the director of the ISSCA since 2016. His published books in Aesthetic Practice and Digital Marketing can be found here. In his presentation this July, he will share his visionary approach to healthcare management and regenerative medicine. Congress attendees will have the opportunity to learn why doctors must use digital marketing strategies to grow a successful practice. Benito Novas will provide tips to attendees on how to capitalize on social media trends to grow practice influence.
- Dr. Maritza Novas, the ISSCA director of education from the USA, will talk about Allogeneic Therapies and Stromal Cell Exosomes. The use of allogeneic therapy is one of the most attractive alternatives to autologous products and is of utmost interest to researchers in recent years. Exosomes serve as mediators for cell-to-cell communication and can be used as cell-free therapeutics for their special characteristics. Dr. Maritza Novas has been in aesthetic and anti-aging medicine since 2001. She is globally recognized for her work in regenerative medicine and dedicated service in education.
- Dr. Silvina Pastrana, the Argentina chapter director of the ISSCA from Argentina, will present on Fundamentals of Cellular Therapies and Mesenchymal Stem Cells (MSC). Dr. Pastrana heads a staff of medical specialists in orthopedics, rheumatology, medical clinic, and cosmetic surgery, performing procedures that incorporate stem cell therapies. She has been serving as a staff surgeon for the Hospital Dr. Prof. Luis Güemes for 21 years.
Besides the above presentations, look forward to sessions in the practical portion of the congress, where the following ISSCA members will show the latest protocols and technologies in regenerative medicine:
- Dr. Julio Ferreira (Argentina), Cosmetic Surgery / Aesthetic Medicine
- Dr. Andrea Lapeire (Argentina), Aesthetic Medicine
- Dr. Maritza Novas (USA), ISSCA Director of Education in the USA
- Dr. Silvina Pastrana (Argentina), Chapter Director of ISSCA in Argentina
The ISSCA members are the leaders in setting standards and promoting excellence in the field of regenerative medicine, related education, certification, research, and publications. The Global Stem Cells Groups (GSCG) is a group of companies, including the ISSCA and other members, dedicated to facilitating stem cell research and medicine. Making the benefits of stem cell medicine a reality for both doctors and patients worldwide is the goal of the GSCG. To learn more, visit our sites at the ISSCA and the GSCG.
- Published in News
Wharton’s Jelly – how does it work?
THURSDAY, 16 SEPTEMBER 2021 / PUBLISHED IN BLOG
What is Wharton’s Jelly?
Wharton’s Jelly is the substance that helps lubricate and support the umbilical cord. What makes Wharton’s Jelly so special is it contains high levels of mesenchymal stem cells. These special stem cells adapt to fit the cells needed to help regenerate damaged tissue and relieve pain naturally.
The Potency of Mesenchymal Stem Cells in Wharton’s Jelly
The mesenchymal stem cells that come from Wharton’s Jelly are among the most potent stem cells, rich in regenerative properties. Wharton’s Jelly also contains a good amount of collagen, hyaluronic acid, and anti-inflammatory properties, making them quintessential in regenerative medicine.
The Role of Regenerative Medicine
Regenerative medicine refers to treatments and medicine that are naturally occurring, including stem cell therapies. Mesenchymal stem cells are harvested from the Wharton Jelly found in the umbilical cord, donated by a healthy mother of a full-term baby. These stem cells contain all the regenerative properties needed to help the body heal and relieve pain naturally.
How Does Wharton’s Jelly Treatment Work?
Mesenchymal stem cells are injected into the body and have the ability to transmute into cells needed to repair damaged tissue and relieve inflammation. These cells harness potent regenerative properties that help rebuild the injured part of the body and provide long-lasting natural pain relief. Unlike pain medicines, which provide temporary relief that masks the symptoms and can be harmful if taken long-term, the stem cells found in Wharton’s Jelly are naturally occurring and natural to the body.
Best Uses for Wharton’s Jelly
Wharton’s Jelly is best used in patients with degenerative diseases and musculoskeletal injuries. The anti-inflammatory and regenerative properties work to repair damaged tissues, leading to lasting pain relief and restored function.
What to Expect from Wharton’s Jelly Treatment
Once a patient and doctor have decided on this type of stem cell treatment, patients can expect a relatively painless process. Stem cells are injected into the site and immediately go to work. Injections are virtually painless, and there are no known negative side effects. Patients can generally get right back to the activities they love. Doctors will discuss treatment plans based on individual needs, and in some cases, resting an injured area for a specific period may be needed.
The Effectiveness of Wharton’s Jelly Therapy
The mesenchymal stem cells found in Wharton’s Jelly have proven to be a very successful therapeutic method to treat several degenerative issues and injuries. This therapy is relatively painless, with no known negative side effects, making it an excellent choice in treatment. Instead of masking the pain with dangerous medicine, stem cell therapy offers a naturally occurring solution that aims to repair the damaged tissue and resolve the problem.
Learn More About Wharton’s Jelly Treatment
If you want to learn more about Wharton’s Jelly Treatment and how you can help your patients, you can check our next training course here: Wharton’s Jelly Training Course.
- Published in Blog
ISSCA Opening of Uccle, Belgium Center with GCell & Stem Cell Training
After several months of organizing and developing a location, the Global Stem Cells Group has finalized the construction of a laboratory and regenerative medicine research center in Uccle, Belgium.
This facility serves both as a regenerative medicine treatment and research center, but also as a plastic surgery clinic. In addition to this, one of the primary goals of the Center is to serve as a place for the fostering of knowledge and experience regarding the different products and equipment required for the application of cellular therapies. As part of the inaugural ceremony a group of physicians were trained in the setup and use of the various regenerative medicine protocols and application process of these cells into the patients.
This center is a push into Europe for the Global Stem Cells Group. As the science advances, newer and more advanced therapies and technologies are being developed and released into the market with each passing month.
With the European Union being one of the largest consumer markets in the world, the finalization of a brick-and-mortar location in Brussels, Belgium is one that will mean great things for the Global Stem Cells Group, and the presence and proliferation of regenerative medicine throughout Europe and the world.
“This was a wonderful opportunity that we could have. It goes to show that even during this pandemic, the medical community is one that is still going strong. We’re still rallying together to look for new breakthroughs, and come up with the most beneficial treatment options for patients,” Said Benito Novas, founder of the International Society for Stem Cell Applications, “In addition to this, it also provides an excellent platform for the distribution of regenerative medicine therapies and equipment, including the revolutionary new GCell Machine,”
Indeed, the inaugural training at the Stem Cell Center in Uccle, Belgium was one that covered both of the more traditional methods of stem cell isolation and application– adipose and bone-marrow derived autologous cells, but this training was also one of the first in which the company had the ability to demonstrate the novel GCell Machine. Using a precise system of blades and filters, it is able to process a stem cell sample from adipose tissue in less than half of the time that it would take a physician to do so through traditional means.
This allows the patient to be more comfortable throughout the shorter procedure, as less anesthesia is also required than when operating under traditional means. The GCell is a minimally invasive, portable machine that allows physicians to fully unlock the potential of regenerative medicine as a component in their practice. Using their global network of distributors and licensed vendors, Global Stem Cells Group is working fervently to ensure no interruption in the global availability of the most cutting-edge regenerative medicine products.
- Published in News
How Cellgenic MSCs Revolutionize Regenerative Medicine

Stem Cells are revolutionizing the field of regenerative medicine, due to their intelligence. Once administered into the patient, they are able to identify and target areas of disease and damage. Adimarket’s Mesenchymal Stem Cell Product excretes growth factors, cytokines, and proteins, which all play a key role in the regeneration of tissue. Their anti-inflammatory and immunomodulatory properties mean that it is difficult for them to be rejected by the body. Additionally, they increase blood flow to the vital organs which need it the most.
Many proprietors of MSC products will claim that it is not necessarily important to have a high ratio of viable cells. They claim that it does not matter how many cells are ‘not viable’, or dead, so long as there is a high enough number of viable cells– however, current research has shown that this is not the case. These dead cells are detectable by the immune system, and it is believed that they can create an inflammatory response within the body at the treatment area, which would lower the effectiveness of the regenerative medicine treatments.
This bending of the science is harmful to our industry, which is why knowledgeable purveyors put one thing above all else– consistency. Our cellular concentrations are the same throughout each batch, and we make sure that there is a high ratio of viable cells. All of our samples are independently verified by a third party laboratory, and have been selected for their phenotypic and genotypic profile, characterized for optimum growth and stability. When the proper care is taken, Mesenchymal Stem Cell products have been identified as having the highest output of growth factors and stem cell factors among the current standards of care– as well as properties of angiogenesis, immunomodulation, and the potential for endogenous repair.
Cellgenic has been working for over a decade, constantly reinventing itself and reinforcing the products that we offer with the latest advancements in the field of regenerative medicine. We take every painstaking measure possible to ensure that the cellular samples that our customers use to treat their patients are second to none– this includes the consistent concentrations of our sample, which are the same throughout ensuring that every patient gets the same treatment. We offer the product in 10 million or 30 million live total nucleated cells, where other fabricators would have the same number of total cells. We ensure that every single product that we send out has been tested for low amounts of annexin, which is a cellular protein which serves as a marker for cell death.
All our Mesenchymal Stem Cell products come in 1cc vials cryogenically preserved– they are shipped overnight within the United States, conveniently delivered to your door in the morning. For use, the product is passively thawed between the palms of your hands– and ready to use when your patients are. MSC 10 contains 10 million live cells and is recommended for a single joint, or a small area. However, the MSC pure pro has 30 million live cells,and can be used for larger applications, or for up to three joints in the same patient at the same time.
If you are interested in finding out more about Cellgenic MSCs, you can send an eMail to info@stemcellsgroup.com
- Published in News
Global Stem Cells Group Announces Training in Brussels, Belgium for April 9/10th
The Global Stem Cells Group, a multi-disciplinary community of scientists and physicians that are collaborating to treat diseases and lessen human suffering through the advancement of the field of regenerative medicine has announced plans for a training in Brussels, Belgium on April 9/10th. This course is a long-awaited return to the country of Belgium after months of inactivity in the region brought about by the COVID-19 pandemic. During this time, research proceeded, and shocking discoveries were made that further thrust regenerative medicine into the world’s stage, and as a result the curriculum has been reinforced with the latest in IV therapy and exosome protocols.
This training will be one of the first of 2021, reaffirming the Global Stem Cells Group’s presence in Europe as a key player in the field of regenerative medicine. Members from both Global Stem Cells Group & the International Society for Stem Cell Application’s Brussels Chapter will be there to assist in expanding research for and the practice of regenerative medicine across Europe.
“It’s good to know that the Global pandemic has not been able to stop the progress of business,” Says Benito Novas, founder and CEO of the Global Stem Cells Group, “With reopening happening slowly, but surely, we hope that more and more physicians and clinics will take up the benefits of regenerative medicine.As we continue to train patients with the GCell Machine, ”
This training is intended to teach physicians the value and process behind incorporating regenerative medicine into their own clinical practice. This includes a theoretical portion that goes over basic regenerative medicine biology and its application, but also includes a Hands-On portion in which doctors, in a controlled environment and guided by a team of medical professionals, will have the opportunity to see procedures be performed just a few feet away, and then get the opportunity to try it for themselves.
The Belgium Stem Cell Center is one that is outfitted with the latest technology in regenerative medicine processing, which ensures any students that pass through it receive an education in extraction, isolation, and application of Platelet-Rich Plasma, Adipose, and Bone Marrow-Derived Mesenchymal Stem Cells.
This training will also teach physicians how to utilize the GCell Machine to perform regenerative medicine therapies. GCell is a tissue homogenization device that is revolutionizing the future of regenerative medicine. It is an extremely compact, all-in-one unit that can homogenize and isolate the stem cells from an adipose sample. GCell’s process of homogenizing with a system of precise blades and filtering ensures that the sample can be processed within an hour, with little input from the physician when the machine begins to do its work.
Once the process is over, the end result is a final product that can be administered to patients within an hour after the initial tissue extraction. This is a far cry from the previous, muti-hour long treatments that physicians have grown accustomed to, and the shortened timespan and simplicity of the procedure is something that both doctors and their patients will greatly appreciate.
If you are interested in enrolling in this upcoming event, or to learn more about the different training opportunities available, you can visit us at our training website.
About Global Stem Cells Group
Global Stem Cells Group (GSCG) is a worldwide network that combines seven major medical corporations, each focused on furthering scientific and technological advancements to lead cutting-edge stem cell development, treatments, and training. The united efforts of GSCG’s affiliate companies provide medical practitioners with a one-stop hub for stem cell solutions that adhere to the highest medical standards.
Global Stem Cells Group is a publicly traded company operating under the symbol MSSV. https://finance.yahoo.com/quote/mssv/
- Published in News