Global Stem Cells Group Announces New President of the International Society for Stem Cell Application (ISSCA)
Miami, Florida – Global Stem Cells Group, a leading organization in the field of regenerative medicine, is pleased to announce the appointment of Dr. Salih Yildirim as the new President of the International Society for Stem Cell Application (ISSCA), a division of Global Stem Cells Group. Dr. Yildirim previously held the position of Director of Overseas Operations at ISSCA.
Dr. Salih Yildirim brings a wealth of experience and expertise to his new role as President of ISSCA. Having previously served as the Director of Overseas Operations, Dr. Yildirim has demonstrated his exceptional leadership skills and strategic vision throughout his career. He has played a vital role in expanding ISSCA’s international presence and fostering collaborations with stakeholders worldwide.
In a statement regarding the appointment, Dr. Daeyong Kim, the former President of ISSCA, expressed his confidence in Dr. Yildirim’s ability to lead the organization. Dr. Kim stated, “I think you are the best leader and the most prepared to be the President of ISSCA. Therefore, I think you should be the head of ISSCA.” Dr. Kim will now transition to other support functions within ISSCA.
Benito Novas, head of Public Relations for ISSCA, expressed his admiration for Dr. Salih Yildirim’s professional qualities. Novas said, “I have known and worked with Dr. Salih for more than 10 years, and I am honored to have him as President within the company. During these years, he has proven to be an exceptional leader with a great vision for ISSCA.”
ISSCA’s objectives under the leadership of Dr. Salih Yildirim will include advancing the understanding and application of stem cell therapies, promoting scientific research and collaboration among professionals in the field, and advocating for the ethical and responsible use of regenerative medicine worldwide. Dr. Yildirim’s extensive experience in business development, international relations, and regenerative medicine will undoubtedly contribute to the achievement of these objectives.
About Dr. Salih Yildirim:
Dr. Salih Yildirim holds a bachelor’s and master’s degrees in business administration from Cleveland State University (United States) and a Ph.D. in health management from the University of Health Sciences. He has held various key positions throughout his career, including Director of Overseas Operations at ISSCA. Dr. Yildirim is currently the General Manager of BioTrend Medical International, a biotechnology firm specializing in genetics, molecular biology, and stem cell technologies. He also serves as the chief executive officer of ReGen.IC Clinic, specializing in regenerative medicine and stem cell applications. Additionally, he is a member of the International Society for Stem Cell Application’s Board of Directors and the Global Stem Cell Group’s Board of Directors and International Operations Manager. Dr. Yildirim is also a member of Istanbul Atlas University’s Board of Directors. He is married and a proud father.
About ISSCA
The International Society for Stem Cell Application (ISSCA) is a multidisciplinary community of scientists and physicians who aspire to treat diseases and lessen human suffering through advances in science, technology, and regenerative medicine.
ISSCA updates its members on advances in stem cell research, MSC, exosomes, and regenerative medicine
The ISSCA’s vision is to take a leadership position in promoting excellence and setting standards in the regenerative medicine fields of publication, research, education, training, and certification.
The international community provides a platform for practitioners to interact with scientists and build medical networks necessary for marketing.
As a medical specialty, regenerative medicine standards and certifications are essential, which is why ISSCA offers certification training in cities worldwide. The goal is to encourage more physicians to practice regenerative medicine and make it available to patients nationally and internationally. Incorporated under the Republic of Korea as a non-profit company, the ISSCA is focused on fostering excellence and standards in regenerative medicine.
About Global Stem Cells Group
Global Stem Cells Group is the parent company of six companies that are dedicated to stem cell research, solutions, and technology training. The group was founded in 2012 and combines dedicated researchers, patient educators, and physician trainers with the shared goal of meeting the need for high-end stem cell solutions and treatments.
Given that the group has a singular focus in this field, it is uniquely positioned to become the global leader in cellular medicine. In addition, by bringing together leading professionals in cellular medicine, it can discover issues that the industry faces and focus its research and development in this area. This specialization has, undoubtedly, enabled it to come up with solutions that address some of the significant issues that most stakeholders are facing in the industry.
Global Stem Cells Group is a publicly traded company operating under the symbol MSSV. https://finance.yahoo.com/quote/mssv/
Safe Harbor Statement: Statements in this news release may be “forward-looking statements”. Forward-looking statements include, but are not limited to, statements that express our intentions, beliefs, expectations, strategies, predictions, or any other information relating to our future activities or other future events or conditions. These statements are based on current expectations, estimates, and projections about our business based partly on assumptions made by management. These statements are not guarantees of future performance and involve risks, uncertainties, and assumptions that are difficult to predict. Therefore, actual outcomes and results may and are likely to differ materially from what is expressed or forecasted in forward-looking statements due to numerous factors. Any forward-looking statements speak only as of the date of this news release, and The Global Stem Cells Group undertakes no obligation to update any forward-looking statement to reflect events or circumstances after the date of this news release. This press release does not constitute a public offer of any securities for sale. Any securities offered privately will not be or have not been registered under the Act and may not be offered or sold in the United States absent registration or an applicable exemption from registration requirements.
- Published in News
Global Stem Cells Group (GSCG) and International Society for Stem Cell Application (ISSCA) to Participate in Dubai Stem Cell Congress 2023
MIAMI, Florida, February 15, 2023 – Global Stem Cells Group (GSCG) and its educational division, the International Society for Stem Cell Application (ISSCA), are proud to announce their participation as sponsors and co-organizers of the Dubai Stem Cell Congress 2023. The congress, endorsed by the Hortman Stem Cell Laboratory, will take place on the 27th and 28th of February 2023 at the prestigious Waldorf Astoria on The Palm in Dubai.
The congress will bring together international experts in regenerative medicine and stem cell research from around the world to share their knowledge, experiences, and insights with the academic community and the general public. The event will also provide a platform for patients to share their success stories of stem cell therapy and how it has helped them overcome various life-threatening diseases.
“We are honored to welcome you to the first edition of the Dubai Stem Cell Congress 2023, which will host international experts in the field of Regenerative Medicine & Stem Cell Research from around the world,” said Dr. Fatma Alhashimi, Chairman of Dubai Stem Cell Congress. “We will be showcasing cutting-edge data, stem cell technologies, and advanced applications.”
GSCG will showcase its range of cellular products and equipment, including exosomes for topical and intravenous use, Mesenchymal Stem Cells (MSC), Bone Marrow Kit, PRP Kit, Fat Kit for obtaining Stromal Vascular Fraction (SVF), workstation, and laboratory equipment. ISSCA, on the other hand, will be represented with its training, congresses, conferences, and the fellowship in Cellular Therapies.
“Our mission is to make cellular therapies a reality for both doctors and patients, and we are happy to continue that mission by sharing our knowledge and showcasing our Cellgenic branded Cellular products and equipment at the congress,” said Benito Novas, Managing Director of Global Stem Cells Group and Head of Public Relations for ISSCA.
With over 25 world-class international speakers, including ISSCA-certified speakers, the congress will feature a variety of panel discussions, lectures, and presentations on the latest innovative technologies in stem cell and regenerative medicine.
ISSCA-certified speakers and experts who will be presenting lectures at the congress include the following:
- Erdinç Civelek, a brain surgeon at Gaziosmanpaşa Taksim Hospital for Research and Education in Turkey.
- Dr. Serdar Kabataş, a brain surgeon at Gaziosmanpaşa Taksim Research and Education Hospital in Turkey.
- Abdulmajeed Hamadi, a consultant hematologist and pioneer in transplantation and stem cell therapy with an FRCP designation in Iraq.
- Salih Yildirim, the Director of Overseas Operations of ISSCA and a member of the Board of Directors of the Global Stem Cell Group and Director of International Operations.
- Benito Novas, the Managing Director of Global Stem Cells Group and Head of Public Relations for the International Society for the Application of Stem Cells in the USA.
Among the esteemed speakers at the event are:
Prof. Anil Dhawan, Dr. Frances Verter, Dr. Masayo Takahashi, Dr. Essam Abdelalim, Prof. Shuibing Chen, Prof. John E. Wagner, Mrs. Kim Petrella, Dr. Edward Guindi, Dr. Mazaiah Yaacob, Dr. Mohammed Moulay, Mr. Oliver Papavlassopoulos, Dr. Sean Ng, Dr. Chiara Cugno, Dr. Siti Aminah, Mr. Thomas Moss, Prof. Dr. Tim Schulz, Dr. Luis Saraiva, Mrs. Renata Mihályová, Dr. Srinivasan Periathiruvadi and Dr. Ahmed Foul.
For the first time in the UAE, patients will have the opportunity to share their success stories of stem cell therapy, including the story of Mahra, a UAE national and survivor of Thalassemia, and the world’s first cord blood patient, Matthew Farrow.
Stem cell therapy is the present and future of medicine and holds the “Hope That Brings Life” to many patients in need. The Dubai Stem Cell Congress 2023 is a landmark event that will bring together leading experts, patients, and members of the academic community to share their experiences and knowledge about stem cell therapy and its advancements.
To learn more about the Dubai Stem Cell Congress 2023 and to make a reservation, visit the https://dubaistemcellcongress.com/ website, email info@stemcellsgroup.com, or call +1 305 560 5337.
About Hortman Stem Cell Laboratory
Mr. Darius Curta Managing Director of Hortman Healthcare Investment – Hortman Stem Cell Laboratory is the first state-of-the-art laboratory within the field of Stem Cells and Regenerative Medicine, focused on providing top-notch cord blood banking solutions. Additionally, our institution will be facilitating the first ever clinical trials in stem cell research in UAE. Hortman Stem Cell Laboratory, located at Golden Mile, Palm Jumeirah, consists of GMP Laboratories & ISO Clean Rooms for stem cell isolation, culture & expansion. Such a facility enables us to provide a solid foundation for successfully establishing an awareness and understanding of stem cells within the Middle Eastern Region. Hortman is leading that charge at the forefront of the research community.
About ISSCA:
The International Society for Stem Cell Application (ISSCA) is a multidisciplinary community of scientists and physicians who aspire to treat diseases and lessen human suffering through advances in science, technology, and the practice of regenerative medicine. ISSCA serves its members through advancements made in the specialty of regenerative medicine.
The mission of the International Stem Cell Certification Agency (ISSCA) is to establish itself as a global leader in regenerative medicine certification, education, research, and training.
ISSCA provides certification training in cities worldwide because it recognizes the importance of standards and certifications in regenerative medicine as a medical specialty. To help more people, both locally and globally, as the demand for more doctors interested in and comfortable with regenerative medicine surges. ISSCA’s mission is to advance quality and uniformity in regenerative medicine worldwide.
About Global Stem Cells Group:
The Global Stem Cell Group is a family of several companies focused on stem cell medicine and research. The company uses its network to bring leadership in regenerative medicine training, research, and patient applications.
GSCG’s mission is to allow physicians to present the benefits of stem cell medicine to patients worldwide. The company also partners with policymakers, educators, and regulators to promote regenerative medicine.
Global Stem Cells Group is a publicly traded company operating under the symbol MSSV. https://finance.yahoo.com/quote/mssv/
To learn more about Global Stem Cells Group, Inc.’s companies visit our website www.stemcellsgroup.com or call +1 305 560 5331
Safe Harbor Statement:
Statements in this news release may be “forward-looking statements”. Forward-looking statements include, but are not limited to, statements that express our intentions, beliefs, expectations, strategies, predictions, or any other information relating to our future activities or other future events or conditions. These statements are based on current expectations, estimates, and projections about our business based partly on assumptions made by management. These statements are not guarantees of future performance and involve risks, uncertainties, and assumptions that are difficult to predict. Therefore, actual outcomes and results may and are likely to differ materially from what is expressed or forecasted in forward-looking statements due to numerous factors. Any forward-looking statements speak only as of the date of this news release, and The Global Stem Cells Group undertakes no obligation to update any forward-looking statement to reflect events or circumstances after the date of this news release. This press release does not constitute a public offer of any securities for sale. Any securities offered privately will not be or have not been registered under the Act and may not be offered or sold in the United States absent registration or an applicable exemption from registration requirements.
- Published in News
ISSCA to Conduct Regenerative Medicine Conference in Cancún, México
Miami, FL, December 19, 2022 – The International Association for Stem Cell Application (ISSCA) has announced plans to host Applications of Stem Cell Therapies in Medicine and Aesthetic Surgery, a regenerative medicine conference, in Cancun, Mexico, April 22, 23, and 24, 2023.
Growing knowledge can turn the tide in the clinical and translational spectrum of stem-cell-based research and therapy, and several obstacles must be overcome. ISSCA’s International Conference, champions new ideas to propel the shift from traditional healthcare to regenerative medicine and therapies. The Conference will also emphasize the role of cutting technology and developments in all areas of stem cell research.
April 22nd & 23rd
The Cancun event will feature a panel of internationally renowned experts on stem cells and regenerative medicine, who will offer a two-day rigorous scientific discourse on critical topics in stem cell research and clinical applications. Topics of focus at the conference include: Tissue engineering and regenerative medicine; updates in Stem Cell research Clinical applications of regenerative medicine and cellular therapies; the latest methods of harvest and isolation, and Non-invasive surgical protocols; strategies for successful delivery of cell products; Ethical and regulatory issues in stem cell technology; and taking advantage of the latest technologies to improve marketing and networking.
April 24th
The third day of the conference (April 24th) will involve intensive hands-on training sessions with ISSCA-certified instructors on stem cell therapy’s aesthetic and clinical applications.
Participants will gain experience and access to different technologies, brands, and products for stem cell therapy.
The leading international conference offers an excellent opportunity for medical practitioners and stakeholders to network and learn from the best in the industry.
All practitioners and stakeholders in the medical sector are invited, including:
- Doctors Researchers & innovators,
- Decision-makers and funding agencies,
- Experts seeking collaborative work on stem cell technology,
- Professional bodies, and
- Marketing professionals.
To learn more about the ISSCA Cancun, Mexico conference and to make a reservation, visit the https://www.issca.us/issca-world-conferences/ website, email info@stemcellsgroup.com, or call +1305 560 5337.
About ISSCA
The International Society for Stem Cell Application (ISSCA) is a multidisciplinary community of scientists and physicians who aspire to treat diseases and lessen human suffering through advances in science, technology, and regenerative medicine.
ISSCA updates its members on advances in stem cell research, MSC, exosomes, and regenerative medicine.
The ISSCA’s vision is to take a leadership position in promoting excellence and setting standards in the regenerative medicine fields of publication, research, education, training, and certification.
The international community provides a platform for practitioners to interact with scientists and build medical networks necessary for marketing.
As a medical specialty, regenerative medicine standards and certifications are essential, which is why ISSCA offers certification training in cities worldwide. The goal is to encourage more physicians to practice regenerative medicine and make it available to patients nationally and internationally. Incorporated under the Republic of Korea as a non-profit company, the ISSCA is focused on fostering excellence and standards in regenerative medicine.
About Global Stem Cells Group
Global Stem Cells Group is the parent company of six companies that are dedicated to stem cell research, solutions, and technology training. The group was founded in 2012 and combines dedicated researchers, patient educators, and physician trainers with the shared goal of meeting the need for high-end stem cell solutions and treatments.
Given that the group has a singular focus in this field, it is uniquely positioned to become the global leader in cellular medicine. In addition, by bringing together leading professionals in cellular medicine, it can discover issues that the industry faces and focus its research and development in this area. This specialization has, undoubtedly, enabled it to come up with solutions that address some of the significant issues that most stakeholders are facing in the industry.
Global Stem Cells Group is a publicly traded company operating under the symbol MSSV. https://finance.yahoo.com/quote/mssv/
- Published in News
Announcement of Training in Fort Lauderdale, Florida by The Global Stem Cells Group
Recently, the Global Stem Cells Group announced plans to teach physicians the value and process behind incorporating regenerative medicine into their own clinical practices. MIAMI, 4–5 November 2022—Global Stem Cells Group, a multidisciplinary community of scientists and physicians that are working together to cure diseases and relieve human pain through the advancement of the field of regenerative medicine, announced today a plan of training in Fort Lauderdale on November 4th and 5th. This training aims to equip physicians with the value and knowledge behind incorporating regenerative medicine into their own clinical practice. Benito Novas, CEO of GSCG, is confident that the event will bring together a group of:
- Doctors are seeking training, products, and equipment in regenerative medicine.
- Doctors who care for patients with conditions that can be treated with cell therapies
- Aesthetic doctors.
- Doctors who want to be up to date with the latest technologies and protocols
- Doctors with an interest in new research on stem cells, MSC, Exosomes, and medical networks want to attract more clients.
The training course is intended to cover:
- Hands-On portion: Doctors, in a controlled environment and guided by a team of medical professionals, will have the opportunity to see procedures being performed a few feet away and then get the opportunity to try them for themselves.
- Review of stem cell biology
- Characterization of cells, cell products, cytokines, and growth factors, as well as their capacity for regeneration.
- Laboratory Processes
- Clinical applications
- Product validation
- Practice Management
- Patient acquisition
The International Society for Stem Cell Application (ISSCA) has done intensive research on this topic and the team will take a lead in educating, training, and certification during the event. As a medical speciality, regenerative standards and certification are very important, which is why ISSCA will provide certificates after the training. By the end of the training course, you will understand everything you need to know to add adult stem cellbased procedures to your existing practice. As before, the united efforts of the seven major medical corporations will provide practitioners with a one-stop hub for stem cell solutions that adhere to the highest medical standards. To find more about the courses and to make reservations for this particular event, head on to their website www.issca.us/autologous-miami-november/ .us email info@stemcellsgroup.com or dial +1305 560 5337.
About ISSCA
ISSCA is a multidisciplinary community that brings together scientists and physicians, all of whom aspire to treat diseases and relieve human pain through advances in science, technology, and the practice of regenerative medicine. ISSCA serves its members through innovations made to the speciality of regenerative medicine. The ISSCA’s vision is to take a leadership position in promoting excellence and setting standards in the regenerative medicine fields of publication, training, research, education, and certification.
As a medical speciality, regenerative medicine standards and certifications are very important, which is why ISSCA provides certification training globally. This is because they want to encourage more physicians to practice regenerative medicine and make it accessible to benefit patients both nationally and worldwide. Incorporated under the Republic of Korea as a nonprofit entity, the ISSCA’s main goal is to promote excellence and standards in the field of regenerative medicine. As a physician, missing this chance to book a personalized hands-on training session must cost you dearly. Contact + 1 305 560 5337 to book as early as possible.
About Global Stem Cells Group
Global Stem Cells Group is the parent company of six companies that are dedicated to stem cell research, solutions, and technology training. The group was founded in 2012 and combines dedicated researchers, patient educators, and physician trainers with the shared goal of meeting the need for high-end stem cell solutions and treatments.
Given that the group has a singular focus in this field, it is uniquely positioned to become the global leader in cellular medicine. In addition, by bringing together leading professionals in cellular medicine, it can discover issues that the industry faces and focus its research and development in this area. This specialization has, undoubtedly, enabled it to come up with solutions that address some of the significant issues that most stakeholders are facing in the industry.
Global Stem Cells Group is a publicly traded company operating under the symbol MSSV. https://finance.yahoo.com/quote/mssv/
- Published in News
Global Stem Cells Group Announces the Launch of a New Bone Marrow Harvesting Kit
The Global Stem Cells Group has announced the launch of a new bone marrow harvesting kit in response to a request for effective and accessible solutions to regenerative medicine procedures
Bone marrow is a spongy tissue that is located inside most large bones – it is essential for generating red blood cells (erythrocytes), white blood cells (leukocytes) and platelets (thrombocytes), using a bone marrow aspirate needle to collect the aspirate can help physicians perform different regenerative treatments especially in osteomioarticular conditions.
Increased Stem and Progenitor Cell Concentrations in Marrow Aspiration
According to the company, the new kit for processing and extraction of pure bone marrow has demonstrated its ability to increase stem and progenitor cell concentrations in marrow aspiration compared to traditional needles. It further adds that the device overcomes the limitations of traditional needles that restrict the amount of peripheral blood that would otherwise have been aspirated by forcing aspiration flow through side ports rather than from an open cannula. This setup has been a major issue with traditional needles and often led to the extraction of excessive amounts of bone marrow as well as impure marrow.
“Cellgenic plans with this new device to position itself as a leader in the development and manufacture of single-use medical devices for a number of medical specialties, we have developed a new bone marrow aspiration and biopsy needle that is manual, sterile and disposable, allowing precise procedures to be performed” said Benito Novas C.E.O of Global Stem Cells group
The new system offers the facility for doctors to reposition the needle more precisely at different access points. Precision is vital when trying to access bone marrow from specific sections of the bone.
According to Benito Novas, CEO of Global Stem Cells Group, the product will be launched at a press conference this September in Istanbul, Turkey within the framework of the World Congress of Regenerative Medicine. Additionally, it will be available at retail for doctors and distributors worldwide from October 2022
Buyers will undergo an orientation to use the new kit. It is easy to work with and many are expected to start using it as soon as they purchase it. The stem cells group will offer all the necessary training and support for the doctors during training and thereafter to ensure that the medical practitioners enjoy the full benefits of the device.
Global Stem Cells Group has been on the frontline in offering quality devices for stem cell procedures. It aims to make procedures as simple, risk-free, and efficient as possible for the maximum benefit of patients.
About Global Stem Cells Group
Global Stem Cells Group is the parent company of six companies that are dedicated to stem cell research, solutions, and technology training. The group was founded in 2012 and combines dedicated researchers, patient educators, and physician trainers with the shared goal of meeting the need for high-end stem cell solutions and treatments.
Given that the group has a singular focus in this field, it is uniquely positioned to become the global leader in cellular medicine. In addition, by bringing together leading professionals in cellular medicine, it can discover issues that the industry faces and focus its research and development in this area. This specialization has, undoubtedly, enabled it to come up with solutions that address some of the significant issues that most stakeholders are facing in the industry.
Global Stem Cells Group is a publicly traded company operating under the symbol MSSV. https://finance.yahoo.com/quote/mssv/
- Published in News
Stem Cell “Tattoo” Technology Allows Researchers to Track Cell Implants Non-invasively
TUESDAY, 12 JULY 2016 / PUBLISHED IN BLOG
Introduction to Stem Cell Tattoo Technology
Researchers at the University of Toronto have developed a tracer ink—a “stem cell tattoo”—that provides the ability to monitor stem cells in unprecedented detail after they’re injected. The research findings, titled “Bifunctional Magnetic Silica Nanoparticles for Highly Efficient Human Stem Cell Labeling,” were published in June in the Journal of Magnetic Resonance Imaging. Already emerging as an ideal probe for noninvasive cell tracking, the technology has the potential to revolutionize stem cell research by arming scientists with the ability to visually follow the pathways and effectiveness of stem cell therapies in the body in real time.
Advancements in Stem Cell Tracking
University of Toronto biomedical engineering professor Hai-Ling Margaret Cheng, a biomedical engineer who specializes in medical imaging, says the new technology allows researchers to actually see and track stem cells after they’re injected. Cheng hopes the technique will help expedite the development and use of stem cell therapies.
Development of the Contrast Agent
Working with colleague Xiao-an Zhang, an assistant professor of chemistry at the University of Toronto, Scarborough, Cheng developed a singular chemical compound known as a contrast agent that acts as a tracer. Composed of manganese, an element that naturally occurs in the body, this tracer compound, called MnAMP, bathes stem cells in a green solution, rendering them traceable inside the body under MRI.
Long-term Cell Tracking with Tracer Ink
The contrast agent “ink” first enters a stem cell by penetrating its membrane. Once inside, it stimulates a chemical reaction that prevents it from seeping out of the cell the same way it entered. Previous versions of contrast agents easily escaped cells. By establishing a way to contain the ink within the cell’s walls, the research team achieved the ability to track the cells long term once they are inside the body.
Advantages of the New Technology
According to Cheng, some basic contrast agents are already available for use in humans, but none are capable of tracking cells over a long period of time. Contrast agents work by illuminating the deepest and darkest corners of a person’s internal architecture so they appear clearly under X-rays, computed tomography (CT) scans, and MRIs. An example of a currently used contrasting agent would be the barium sulfate solution given to patients to help diagnose certain disorders of the esophagus, stomach, or intestines.
Non-invasive Cell Tracking Benefits
Before the stem cell tattoo tracer ink was developed, surgery was the only option for scientists to get a literal glance of a cell’s destiny after it was injected into the body. Now, researchers can track the results in real time without resorting to any invasive procedures. “Before, we could not visually track the cells once they were introduced into the body,” Cheng says. “Now we have the ability to view cells in a non-invasive manner using MRI, and monitor them for potentially a very long time.”
Current Stage of Development

Currently, the tracer ink technology is still in the early development phase and requires more animal testing. Cheng is hopeful it can proceed to human clinical trials in about 10 years. While Cheng has already proven that tattooing an animal’s embryonic stem cell doesn’t affect its ability to transform into a functional heart cell, larger models, such as rats or pigs (which better represent human size), are up for evaluation next.
Future Testing and Applications
In those test cases, researchers will cut off and reduce blood flow in the animals to mimic the effects of damage caused by a human heart attack. Cardiac stem cells pre-tagged with Cheng’s ink tracer technology will then be injected into the damaged tissue. Using MRI to monitor the luminous inked stem cells in action, researchers can non-invasively follow where in the body they’re traveling and more easily determine if the new cells are responsible for restoring normal heart rhythm. Before it can be tested in humans, the chemical tracer will also have to pass rigorous toxicology tests to ensure its safety.
- Published in Blog
Could stem cells offer the cure for muscular dystrophy?
Exploring Stem Cell Therapies for Muscular Dystrophy
Muscular dystrophy (MD) encompasses a group of genetic disorders causing progressive muscle weakness and loss of mass. At Global Stem Cells Group, we delve into the potential of stem cell treatments to revolutionize care for muscular dystrophy patients worldwide.
Understanding Muscular Dystrophy
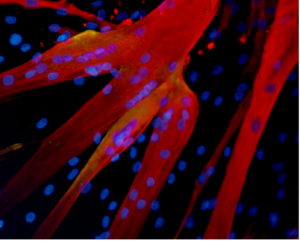
Muscular dystrophy affects voluntary muscles, with symptoms ranging from mild to severe, impacting mobility and daily life. Duchenne muscular dystrophy (DMD), the most severe form, primarily affects young boys, leading to progressive muscle degeneration and shortened lifespan.
Challenges in Current Treatments
While treatments like physiotherapy and steroids aim to manage symptoms, there is no cure for DMD. These approaches often fall short due to side effects and limited effectiveness in halting disease progression.
The Promise of Stem Cells in Muscular Dystrophy
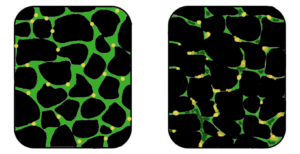
Stem cell therapies offer potential by addressing the underlying cause—defective dystrophin production. Research explores using stem cells from muscle tissue, bone marrow, and blood vessels to regenerate muscle fibers deficient in dystrophin, showcasing promising results in animal models.
Recent Advances and Research
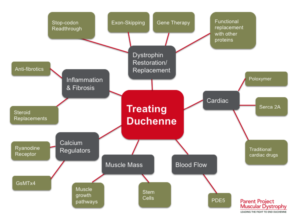
Studies have demonstrated that stem cells isolated from muscle blood vessels can restore mobility in affected animals. Researchers have also explored genetic correction combined with stem cell therapy, showing improvements in muscle regeneration and function in mice models of DMD.
Future Directions
While translating these findings to human treatments requires further research, combined therapies integrating stem cells and gene therapies show promise. These approaches aim to replace defective stem cells with healthy ones or reduce inflammation to preserve muscle function effectively.
Conclusion
While a cure for muscular dystrophy remains elusive, stem cell research holds immense promise. Global Stem Cells Group remains at the forefront, exploring innovative therapies that could transform the outlook for patients with muscular dystrophy.
References:
- http://www.mayoclinic.org/diseases-conditions/muscular-dystrophy/basics/definition/con-20021240
- http://www.eurostemcell.org/factsheet/muscular-dystrophy-how-could-stem-cells-help
- https://www.mda.org/disease/duchenne-muscular-dystrophy/research
- http://quest.mda.org/article/scientists-bullish-stem-cells-muscle-repair
- http://hsci.harvard.edu/stem-cells-used-treat-muscular-dystrophy-mice
- https://med.stanford.edu/news/all-news/2014/12/stem-cells-faulty-in-duchenne-muscular-dystrophy.html
http://www.mayoclinic.org/diseases-conditions/muscular-dystrophy/basics/definition/con-20021240
- http://www.eurostemcell.org/factsheet/muscular-dystrophy-how-could-stem-cells-help
- https://www.mda.org/disease/duchenne-muscular-dystrophy/research
- http://quest.mda.org/article/scientists-bullish-stem-cells-muscle-repair
- http://hsci.harvard.edu/stem-cells-used-treat-muscular-dystrophy-mice
- https://med.stanford.edu/news/all-news/2014/12/stem-cells-faulty-in-duchenne-muscular-dystrophy.html
- Published in Blog







