EXOSOMES
One major aims of regenerative medicine is to replace lost tissue with new cellular material or to improve the regeneration of damaged, malfunctioning, diseases tissue and organs using stem cell transplantation. In view of this, the discovery of Extracellular vesicles (EVs) in the twentieth century have being considered as significant factors in inflammation and immune responses, antigen presentation, immunomodulation, coagulation, tissue regeneration, organ repair, cell-cell communication, senescence, proliferation and differentiation in the body [1]. Extracellular vesicles are believed to be involved in many biological processes and they can be modified to contain specific proteins, genetic lipids, and genetic materials including messenger RNA (mRNA), microRNA (mRNA), and other small non-coding RNAs, and genomic DNA (gDNA) from their progenitor cell.
Extracellular vesicles are classified into two groups which include; exosomes and ectosomes. Exosomes are characterized with cup-shaped morphology, appearred as flattened spheres with diameters ranging from 30 to 150 nm. Similarly, exosomes have a characteristic lipid bilayer which has an average thickness of ∼5 nm. Thus, the lipid components of exosomes include ceramide (sometimes used to differentiate exosomes from lysosomes), cholesterol, sphingolipids, and phosphoglycerides with long and saturated fatty-acyl chains. The exosome is formed or derived from a multivesicular body (MVB) when the MVB fuses with the plasma membrane and is released into the extracellular environment.
Diagrammatic representation of a medium size Exosome.
Exosomes contain many types of biomolecule, including proteins, carbohydrates, lipids and nucleic acids which vary depending on the EV’s origin, its physiological and pathological state, and even the precise cellular release site. Thus, the protein composition within can also mark the existence of disease pathologies such as inflammatory diseases; however, exosomes also contain a number of common proteins as well as those that participate in vesicle formation and trafficking. Furthernore, exosome plays a role in intercellular communication by carrying proteins and RNAs between neighboring cells or even to distant organs, they also bind to cell membranes through receptor– ligand interaction and mediate antigen presentation, cancer progression etc.
Various techniques have being developed for the isolation of exosomes which includes; ultracentrifugation-based isolation techniques, size-based techniques, precipitation techniques, immune-affinity capture-based techniques, and some novel combination techniques. Exosomes primarily exist in pellets after centrifugation 100000–200000×g and the use of Ultracentrifugation and ultrafiltration can be used to obtain purified exosomes in the laboratory, but this technology is difficult to apply on a large scale.
Exosomes can be stored at 4°C for up to 1 week for short-term while it can be stored at -80°C for long-term by suspending it in phosphate buffered saline.
More importantly, growing evidence has also suggested that exosomes play a key role in facilitating tumorigenesis by regulating angiogenesis, immunity, and metastasis. Circulating exosomes in body fluids and blood in particular are potentially non-invasive or minimally invasive biomarkers for early diagnosis and prognosis of various types of cancer.[6] As the first step towards improving human health, exosomes have to be reliably and efficiently isolated from complex biological matrices like blood, urine, and cerebrospinal fluid since they are currently tested as next-generation biomarkers in those body fluids.
In conclusion, it is advantageous to use exosomes for cell based treatments because the use of exosomes can avoid problems associated with the transfer of cells, which may already have damaged or mutated DNA. Also, exosomes are small and can easily circulate through capillaries, whereas the cells used in other cell-based therapies, such as MSCs, are too large to go through capillaries, and thus cannot get beyond first pass capillary beds, such as the lungs.
The level of MSCs in cell-based therapies may quickly diminish after transplant while, exosomes can achieve a higher “dose” than the transplanted MSCs. Similarly, exosomes can also be utilized to tackle toxicity and immunogenicity problems resulting from such biomaterial treatments as nanoparticles.
CLINICAL APPLICATION
Ø Immuno-modulatory and anti-inflammatory properties of Exosomes could be the potential biological mechanisms for clinical treatment to promote bone regeneration.
Ø Adipose-derived stem cell-derived exosomes promote fracture healing in animals by binding to polylactic acid-glycolic acid scaffolds.
Ø Exosome can be used for the treatment of chronic kidney disease, type 1 diabetes mellitus, and skin damage.
Ø MSCs-derived exosomes have shown therapeutical benefits in stroke and intravenous administration of MSCs-derived exosomes induced an increase of neurogenesis, neurite remodeling, and angiogenesis.
Ø Administration of MSCs-derived exosomes’ has being observed in a traumatic brain injury model by showing an inflammation reduction and good outcomes.
Ø The injection of MSCs-derived exosomes has also been shown to be a possible treatment for spinal cord injury (SCI), by reducing inflammation and by promoting neuro-regeneration in rats after injury.
Ø Exosomes can be used as a delivery system of therapeutic signals or drugs due to their low immunogenicity, ability to cross the blood-brain barrier (BBB), and long half-life in circulation.
- Published in Blog
What Are The Best Options For Managing Chronic Pain?
Chronic pain. Roughly translated as a condition of persistent misery for people. It is a serious health condition that has both physical and psychological suffering and is often associated with a specific ailment like arthritis, migraine, frozen limb, etc. People who suffer from chronic pain perceive themselves to be in a state of constant agony and distress, which can affect their ability to lead a ‘normal’ life, from being able to go to work every day to social interaction.
There are several treatment options that people resort to for managing persistent pain conditions. In this article, we talk about those treatment options with the stem cell regeneration being the most current and promising one. Also, we will discuss the potential benefits and pros and cons of regenerative medicine.
What is chronic pain and how is it caused?
During ‘PAINWeek2017’ in Las Vegas, Dr. Jay Joshi, MD, explained the connection between inflammation and pain. He states that inflammation is triggered by various chemical and physical stimuli, and is a normal phenomenon of the body to protect the injured area and speed up the healing process. When inflammation exceeds its normal extent, it conversely delays the healing process and forms this cycle of non-resolving inflammation that leads to chronic pain.
Chronic pain is a multifactorial condition. It can be caused by medical harm, injury or nerve damage. However, it can also be caused by environmental factors, for example, back pain due to incorrect posture, or physiological factors such as aging, weak bones.
Common conditions that are related to chronic pain are:
- Osteoarthritis: the degeneration of cartilage in the joint and the corresponding bones, leading to pain and stiffness mainly in thumb, knee and hip joints.
- Migraine: a half-sided recurrent headache that gives a throbbing sensation and is accompanied by blurred vision and nausea.
- Multiple Sclerosis: a chronic, progressive condition that involves nerve cell sheath damage in the spinal cord and brain. Its symptoms include severe fatigue, speech disturbances, blurred vision and numbness.
- Neuropathy: a condition that involves damage to the peripheral nerves causing weakness, pain and numbness, particularly in the hands and feet.
- Fibromyalgia: a soft tissue disorder that involves widespread musculoskeletal pain also characterized by fatigue, sleep disturbance and mood swings.
According to a 2016 analysis by the National Health Interview Survey, approximately 50 million Americans suffer from chronic pain. It is considered to be one of the most common conditions that medical treatment is sought out for. Some of the repercussions of chronic pain that agonize people are:
- Depression and mood swings
- Poor performance
- Poor social life
- Restricted mobility
- Anxiety
- Poor self-perception about life
- Dependence on opioids
Although it is necessary to diagnose and treat the underlying cause of pain, doctors deal with pain as a separate entity and provide symptomatic treatments to save patients from the continuous discomfort it brings.
Management of Chronic Pain
Some of the common management strategies adopted by doctors are:
Analgesics
There are several over the counter pain killers available that are widely used by patients with chronic pain. These include paracetamol, acetaminophen, non-steroidal anti-inflammatory drugs (NSAIDs), aspirin, ibuprofen, naproxen, etc.
Although analgesics are considered safe, long term usage can produce a number of side effects. It can lead to liver damage and is specifically dangerous for patients with liver cirrhosis. Likewise, NSAIDs are known to affect the kidneys and increase the risk of a heart attack.
Anticonvulsants
Drugs that are originally used to treat epilepsy can also be an effective management strategy against chronic pain. For example, the second-generation anticonvulsants Gabapentin and Lyrica are approved by FDA for the treatment of pain. First-generation anticonvulsants such as carbamazepine and phenytoin are also considered effective. However, these first-generation drugs have more side effects when compared to second-generation drugs. They can cause side effects like ataxia, liver damage, sedation, etc.
Antidepressants
Doctors also prescribe antidepressants for the treatment of pain. These comprise of tricyclics like amitriptyline, imipramine or doxepin. These drugs can be given to patients with or without depression as it has distinct effects of pain relief and mood elevation.
Similarly, pain in fibromyalgia and diabetic nerve pain are treated with other types of antidepressants – serotonin and norepinephrine reuptake inhibitors (SNRIs) such as venlafaxine and duloxetine. These are equally effective as tricyclics but have comparatively milder side effects.
Opioids
Opioids such as codeine, morphine, oxycodone, etc. are very effective against severe chronic pain conditions. However, these drugs are not available over the counter and not provided by the pharmacist without prescription because of their risk of addiction.
Nonetheless, experts state that if the dose is vigilantly managed, their potential benefits can outweigh the risks. That is why doctors carefully observe patients prescribed with opioids for chronic pain to avoid any untoward dependence.
Alternative medicine
If traditional medicine hasn’t worked, many people often resort to other naturally occurring remedies for its cure. Alternative medicine varies from herbal remedies and therapies that are naturally available in plants that have curative properties. Cannabinoids, Akuamma and Kratom plants are a few examples with such characteristics that are currently trending.
Could Stem Cell Regeneration (SCR) be the answer?
As the human central nervous system matures and becomes more complex, the regenerative ability of the body further reduces. Regenerative medicine caters for this by focusing on the 3 Rs; Replacement, Repair, and Restoration of tissues and their function. This can be directed for the treatment of non-resolving inflammation by generating ex vitro cells, tissues or organs which can then be transplanted into the body.
Stem cell therapy is a unique model of treatment that replaces injured or lost cells, which can help 1.5 billion people that are experiencing chronic pain worldwide, with 23-26% of the people suffering from lower backache alone. According to Dr. Jay in ‘PAINWeek2017’, there are two types of stem cell regenerative therapy: Autologous and Non-Autologous.
In autologous SCR, mesenchymal stem cells (MSCs) are derived from adipose tissue of the patient and transplanted back into the injured area, where they potentially differentiate into the respective cells. For the other type, the MSCs are derived from embryonic tissues.
Research published in Pain Physician Journal in 2017 shows that MSC therapy is a great alternative because they are readily available. In addition, their homing potential is also considered a pro and they can be grown using standard culture techniques. It has also shown that autologous SCR is potentially safe for both humans and animals.
There has been a reported reduction from pre to post-operative pain with the use of SCR. However, various complications have been acknowledged in studies. Firstly, there’s a risk of tumor formation at the site of transplantation. Secondly, there have been instances of undesired bone formation. Furthermore, there is also a risk of abnormal immune reactions to occur.
The effective results of cell-based therapy are promising, with evident results in the treatment of chronic pain conditions in osteoarthritis, degenerative disc disease as well as neuropathic pain. However, there is still a need for advanced clinical research to deepen the understanding of the mode of action of MSCs and their potential outcomes inside the human body.
- Published in Blog
MESENCHYMAL STEM CELLS
Over the last decade, cellular therapy has developed quickly at the level of in vitro and in vivo preclinical research and in clinical trials. Thus, one of the types of adult stem cells that have provided a great amount of interest in the field of regenerative medicine due to their unique biological properties is Mesenchymal stem cells (MSCs).
Mesenchymal stem cells (MSCs) are known to be multipotent stromal cells that can differentiate into a variety of cell types which include osteoblasts (bone cells), chondrocytes (cartilage cells), myocytes (muscle cells) and adipocytes (fat cells which give rise to marrow adipose tissue). Furthermore, MSCs are responsible for tissue repair, growth, wound healing and cell substitution resulting from physiological or pathological causes; they have various therapeutic applications such as in the treatment of central nervous system afflictions like spinal cord lesions (1). Similarly, they are characterized by an extensive capacity for self-renewal, proliferation, potential to differentiate into multiple lineages and their immune-modulatory role on various cells.
Mesenchymal stem cells have the ability to expand in many folds in culture while retaining their growth and multilineage potential. Also, they can be identified by the expression of many molecules including CD105 (SH2) CD73 and CD34, CD45. Thus, these properties of MSCs make these cells potentially ideal candidates for tissue technology.
In addition, it has been discovered that MSCs, when transplanted systemically, have the ability to transport to sites of physical harm or damage in animals, suggesting that MSCs have migratory capacity. This migration property of MSCs is important in regenerative medicine, where various injection routes are utilized depending on the damaged tissue or organ.
The first source of Mesenchymal stem cells was in the bone marrow and considered to be the gold standard for clinical research, although various other sources have being discovered which include: Adipose tissue, Dental pulp, Mobilised Peripheral blood, Amniotic fluid, Joint synovium, synovial fluid, Endosteum, Periosteum, Menstrual blood and birth-derived tissues.
Cohnheim (German Biologist) hypothesized in the late nineteenth century that fibroblastic cells derived from bone marrow were involved in wound healing throughout the body, while in 1970, Alexander Friedenstein described a population of plastic-adherent cells that emerged from long-term cultures of bone marrow and other blood-forming organs, and he showed to have colony forming capacity and osteogenic differentiation characteristics in vitro as well as in vivo upon re-transplantation.
Arnold Caplan (1991), coined the term “mesenchymal stem cell and stated that the cells as multipotent mesenchymal cell populations which can differentiate into several tissue types, and demonstrated roles for MSCs in the regeneration of bone, cartilage or ligaments in animal and clinical studies. However, the first clinical trials of MSCs were completed in 1995 when a group of 15 patients were injected with cultured MSCs to test the safety of the treatment.
According to International Society for Cellular Therapy, the proposed minimum criteria to define MSCs include the following:
(a) The cells should exhibits plastic adherence
(b) The cell should possess specific set of cell surface markers, i.e. cluster of differentiation (CD) 73, D90, CD105 and lack expression of CD14, CD34, CD45 and human leucocyte antigen-DR (HLA-DR).
(c) The cells should have the ability to differentiate in vitro into adipocyte, chondrocyte and osteoblast.
Thus, these characteristics are valid for all MSCs, although few differences exist in MSCs isolated from various tissue origins.
Mesenchymal stem/stromal cells (MSCs) can be isolated from neonatal tissues, most of which are discarded after birth, including placental tissues, fetal membranes, umbilical cord, and amniotic fluid. Placenta is an ideal starting material for the large-scale manufacture of multiple cell doses of allogeneic MSC. The placenta is a fetomaternal organ from which either fetal or maternal tissue can be isolated.
MSC derived from placenta have long-term proliferation and immunomodulatory capacity, superior to bone marrow-derived MSC. The placenta is a fetomaternal organ consisting of both fetal and maternal tissue, and thus MSC of fetal or maternal origin can be, theoretically isolated. Thus, neonatal tissues are easily available and they have biological advantages in comparison to adult sources that make them a useful source for stem cells including MSCs. They appear to be more primitive and have greater multipotentiality than their adult counterparts.
Clinical Application
Ø MSCs have been widely used to treat immune-based disorders, such as Crohn’s disease, rheumatoid arthritis, diabetes, and multiple sclerosis.
Ø MSCs have been widely used as a treatment for numerous orthopedic diseases, including bone defects, osteoarthritis (OA), femoral head necrosis, degenerative disc, spinal cord injury, knee varus, osteogenesis imperfecta, and other systemic bone diseases.
Ø MSCs are promising cell source for treatment of autoimmune, degenerative and inflammatory diseases due to the homing ability, multilineage potential, secretion of anti-inflammatory molecules and immunoregulatory effects.
Ø MSCs play a key role in the maintenance of bone marrow homeostasis and regulate the maturation of both hematopoietic and non-hematopoietic cells.
Ø MSCs have been shown to be powerful tools in gene therapies, and can be effectively transduced with viral vectors containing a therapeutic gene, as well as with cDNA for specific proteins, expression of which is desired in a patient.
Ø It has been proved that MSCs can differentiate into insulin producing cells and have the capacity to regulate the immunomodulatory effects
REFERENCES
- Wei X, Yang X, Han ZP, Qu FF, Shao L, Shi YF. Mesenchymal stem cells: a new trend for cell therapy. Acta Pharmacol Sin. 2013 Jun; 34(6):747-54
- Friedenstein AJ, Piatetzky II S, Petrakova KV. Osteogenesis in transplants of bone marrow cells. J Embryol Exp Morphol. 1966;16:381–90
- Friedenstein AJ, Chailakhyan RK, Latsinik NV, Panasyuk AF, Keiliss-Borok IV. Stromal cells responsible for transferring the microenvironment of the hemopoietic tissues. Cloning in vitro and retransplantation in vivo. Transplantation. 1974;17:331–40.
- Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, Mosca JD, et al. Multilineage potential of adult human mesenchymal stem cells. Science. 1999;284:143–7.
- Caplan AI, Dennis JE. Mesenchymal stem cells as trophic mediators. J Cell Biochem. 2006;98:1076–84.
- Wang S, Qu X, Zhao RC. Clinical applications of mesenchymal stem cells. Journal ofHematology & Oncology. Apr 30, 2012;5:19.
- Dominici, M., Le Blanc, K., Mueller, I., Slaper-Cortenbach, I., Marini, F., Krause, D., Deans, R., Keating, A., Prockop, D. and Horwitz, E. (2006) Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular. Therapy position statement. Cytotherapy 8, 315–31
- Published in Blog
Which are the best options For Treating Common Sports Injuries?
As physicians, we are constantly dealing with sports injuries. While they are typically not seen as life-threatening with high chances of recovery, they could potentially cause further problems down the line for the patient. Sports injuries can also take months to heal and getting patients back on their feet can take a lot of time, effort and money, which can be frustrating for patients who want to get back to playing football every weekend or for those who have a skiing holiday booked in the next month.
Recently, there has been a massive surge in the use of stem cells as an alternative treatment to common sports injuries. This article aims to outline the benefits and risks attached to using stem cells and how this alternative treatment may help patients who are suffering from common sports injuries.
What Are The Most Common Sports Injuries?
We are constantly treating sports injuries on a regular basis. Some of the most common sports injuries doctors typically deal with are:
Ankle Sprain
An ankle sprain occurs when the ligaments in the ankle are stretched and eventually torn due to twisting or falling onto the foot. Ankle sprains are common among athletes and sports enthusiasts, however, if left untreated, the ankle can weaken, making it more vulnerable to further damage. This leads to long term problems such as chronic ankle pain, arthritis, and ongoing instability.
A sprained ankle can be easily diagnosed when we see displays of swelling, bruising, stiffness and pain when we attempt to touch or move the ankle.
Groin Pull
A groin pull is basically a groin strain. Strains occur when the muscles are overstretched, moving in directions that are not normal for them or pulled too forcefully or suddenly. This leaves them torn and damaged and results in tenderness and bruising in the groin and inner thigh area. This injury is common in athletes that play sports which require a lot of quick side-to-side movements.
A groin pull can be easily diagnosed after a thorough physical examination of the symptoms and possible tests such as x-rays and MRI’s.
Hamstring Strain
A hamstring strain occurs when the three muscles in the back of the thigh are overstretched from movements such as hurdling and kicking the leg out sharply. As these muscles are naturally tight and susceptible to sprains, it can take from six to twelve months to heal and are vulnerable to recurring injuries. Poor or lack of stretching are the likely causes of a pulled hamstring.
Shin Splints
Shin Splints is an inflammation of the muscles in the lower leg when they are overworked and stressed. Shin splints are often found in athletes that engage in sports that require a lot of running, dodging or quick stops and starts.
Knee injury: ACL tear
An ACL knee injury is a tear or sprain of the ligament that holds the leg bone to the knee. Sudden stops or changes in direction can tear the ACL and make that dreaded “pop” sound. Almost immediately, the knee will swell, feel unstable and be too painful to bear weight. This injury is common in athletes that engage in sports such as soccer, basketball, and downhill skiing.
ACL tears are commonly seen as a severe sports injury and can be traditionally treated with rehabilitation programs and surgery to strengthen or completely replace the torn ligament.
Knee injury: Patellofemoral Syndrome
Patellofemoral Syndrome occurs when repetitive movements of the kneecap (patella) are made against the thigh bone (femur) which damages the tissue. This knee pain is common among young adults and can be caused by a number of factors such as weakness in the thigh or buttock muscles, tight hamstrings, short ligaments around the kneecap or alignment problems through the feet.
Tennis Elbow (epicondylitis)
Tennis Elbow is common for athletes that play sports such as tennis or golf that require the player to ‘grip’ tightly and repetitively for an extended amount of time. This results in the ligaments of the forearm becoming strained and inflamed, making it painful to make wrist or hand motions.
What Are The Traditional Treatment Options & Management Strategies For Sports Injuries?
Traditional treatment methods and strategies for treating mild sports injuries such as sprains and strains can be done at home. Traditional advice given by doctors can include taking rest, applying ice to reduce the swelling and dressing the injury with compression bandages to support and assist healing. Nonsteroidal anti-inflammatory drugs (NSAIDs) such as ibuprofen may also be prescribed to help reduce swelling and to relieve pain.
Moderate severity injuries may be traditionally treated with immobilization strategies such as the use of crutches or a cast brace to assist the ankle to heal for the first few weeks. This may be followed with rehabilitation exercises to strengthen the ankle and prevent stiffness and future problems.
Severe sports injuries such as knee injuries are traditionally treated with surgery or rehabilitation programs to replace or restore the torn ligaments or muscles. This also goes for those patients who experience persistent problems after months of nonsurgical treatment.
Whatever the severity level of the sports injury, the healing process can take up to several months before the muscle restores to its natural condition. Injuries can also leave muscles and ligaments permanently weakened and susceptible to further injury.
Is Stem Cell Therapy An Option For Treating Sports Injuries?
The use of stem cells to treat sports injuries are becoming more popular due to its ability to grow new blood vessels that facilitate faster and better healing, decrease or prevent inflammation and release proteins (cytokines) that can slow down tissue degeneration and reduce pain. Sports injuries that include damage to tendons, ligaments, muscles, and cartilage are reported to be seeing the best results from stem cell therapy.
Dr. Bill Johnson, MD of Innovations Medical sees stem cell therapy as a revolutionary alternative to painful surgery and long recovery and states that,
“Patients who undergo stem cell therapy for their sports injury report a reduction in their painful symptoms and increase range of motion and increased mobility. Stem cell therapy helps to quickly reduce joint inflammation, and many patients see improvements in 1 to 2 days. Anti-inflammatory results of the procedure can last for 2 to 3 months and many patients see a gradual improvement in their condition over time.”
Even celebrity athletes such as Peyton Manning and Ryan Tannehill have used stem cell treatment in conjunction with traditional treatments with success. Stem cells are placed directly into the joint via surgical application or injection to aid quicker healing and promote the growth of cells needed to restore strength and flexibility in muscles and ligaments.
However, using stem cells as an alternative method to treat sports injuries is still a controversial subject and is very much still debated among medical professionals. Research is still undergoing to show whether or not stem cell therapy is the best solution. Critics of this therapy argue the fact that stem cell therapy doesn’t work any better than a placebo and that there is no clear evidence that this type of therapy is safe. Unwanted side effects can include swelling and pain and if stem cells are used from other sources or manipulated in any way, this can result in a higher risk of developing tumors.
- Published in Blog
How To Effectively Treat Amyotrophic Lateral Sclerosis (ALS)
Although a rare neurologic condition, Amyotrophic Lateral Sclerosis (ALS) is the most common type of Motor Neuron Disease (MND), a condition that affects the voluntary muscles. This is a progressive disorder that leads to muscle weakness and depletion due to nerve dysfunction.
ALS is also called Lou Gehrig’s disease, named after the football player who had this condition. The literal meaning of Amyotrophic is ‘no muscle nourishment’ which becomes the cause of muscle atrophy. ‘Lateral’ refers to the group of nerves in the spinal cord that sends signals to the muscles. It is these nerves that degenerate, leading to sclerosis in this region. In later stages, this affects the nerves that control breathing and hence can be fatal.
The initial symptoms of ALS include stiffness and muscle weakness, which gradually involves all the muscles under voluntary control. The affected regions and progressive pattern vary from one person to another. Some having difficulty holding a pen or a cup while others finding difficulty speaking, chewing or even talking. Thus, ALS is an ailment that affects daily life and makes simple tasks painful and troublesome.
According to the Center for Disease Control and Prevention (CDC), 14,500 to 15,000 people had ALS in the United States in 2016, with approximately 5000 people having a confirmed diagnosis for the condition annually. Although the average survival rate is three to five years, patients can live for ten years or more.
Are there different types of ALS?
Amyotrophic Lateral Sclerosis has two types:
Sporadic ALS: this is the most common one and affects 95% of sufferers. This type occurs without a clear cause.
Familial ALS (FALS): This type occurs in 5-10% of sufferers. This type of ALS is genetic and runs in families. This occurs due to abnormal changes to a gene that is then passed in generations.
What are the symptoms of ALS?
Early signs and symptoms might be unnoticeable and become perceptible after some time. Most clinical signs are evident of upper motor neuron and lower motor neuron lesion. The limb onset ALS (70%) involves initial symptoms in the limbs while the bulbar onset ALS (25%) is characterized by speech and swallowing problems. This is followed by weakness in the limbs later. The remaining 5% of the patients have respiratory involvement in the early period. [5]
Most common symptoms include:
- Muscle weakness in the limbs (distal or proximal)
- Asymmetric progressive muscle wasting
- Difficulty in motor activities like walking, talking, chewing
- Weakness in arms, legs, hands, and feet
- Muscle cramps and twitching
- Slurred speech
- Fatigue
- Emotional liability (episodes of uncontrolled laughing and crying)
- Difficulty in maintaining posture and gait
- Difficulty in breathing and swallowing
With the progression of the disease, symptoms may spread to all parts of the body. In some patients, frontotemporal dementia may occur resulting in poor memory and decision-making abilities.
What Causes ALS?
The exact cause of ALS has not been known by scientists to date. However, research is being carried out to understand what causes ALS. There are several different factors such as:
Genetic changes
Studies have shown that 5 to 10% of cases of ALS are caused by genetic mutations. For example, changes to the gene that makes SOD1 protein causes damage to motor neurons.
Environmental factors
No major association has been established between environmental factors like toxins, viruses, diet or physical trauma and the risk of development of ALS. However, there is ongoing research on the subject. Studies have shown that some athletes are at a higher risk of acquiring ALS due to vigorous physical activity.
Chemical disturbance
Glutamate is the neurotransmitter that is in control of signals to and from the brain. Accumulation of this neurotransmitter within the spaces surrounding the nerves damages them.
Research has also shown mitochondrial structural and functional abnormalities, as well as defects in axonal structure and transport, could be the causative agents for ALS.
How Do We Diagnosis ALS?
When it comes to diagnosis, there are no specific tests that can provide a definitive diagnosis for ALS. However, doctors conduct a series of tests to rule other similar diseases. A full medical history check and a neurologic examination are undertaken at regular intervals to assess the progressive worsening of symptoms.
Running the following Image testing diagnostic tests can help reach a diagnosis:
- Electromyography (EMG) – EMG records the electrical activity of the muscle fibers.
- Nerve Conduction Study (NCS) – NCS assesses the electrical activity of the nerves and muscles.
- Magnetic Resonance Imaging (MRI) – MRI rules out other possible conditions such as a tumor or cyst in the spinal cord, cervical spondylosis, or a hernia in the neck that could be causing the nerve compression.
Laboratory tests such as blood screening and urine tests can also be carried out so that other diseases can be eliminated.
What Are The Treatment Options & Management Strategies For ALS?
ALS is managed through a multidisciplinary approach.
Unfortunately, there is no definitive cure for the disease at this time. Management of ALS is done through symptomatic treatment to ease the condition of the patients and prevent unnecessary complications:
Support – Physicians, psychologists, speech therapists, nutritionists, and home care assistance all play a vital role in making life easier for patients with ALS.
Medication – Riluzole (Rilutek) and Edaravone (Radicava) are the drugs approved by the U.S Food and Drug Administration (FDA) for treating ALS. Riluzole is believed to reduce glutamate levels thereby, decreasing damage to the motor neurons. Edaravone acts an antioxidant and is believed to expel free radicals and reduce the oxidative stress in the motor neurons.
Lifestyle Habits – Physiotherapists can recommend exercise and physical activity like walking, swimming, and bicycling that may improve muscle strength and help elevate mood without overstressing the muscles.
Speech Therapy – therapists can help patients with ALS to employ strategies to speak clearly. They may also recommend computerized aids such as speech synthesizers and eye-tracking technology to help people learn ways for responding by nonverbal means.
Diet – Nutritionists may formulate a diet plan for patients, which consists of food that is easy to swallow and provides enough nourishment and calories for the patients to maintain adequate energy levels and to prevent excessive weight loss
Breathing Support – Patients with ALS may suffer from shortness of breath and difficulty breathing during physical activity or while lying down. If this is the case, doctors can recommend Non-Invasive Ventilation (NIV) that provides breathing support through the nose or mouth. NIV improves quality of life and increases the number of years of survival for patients.
Is Stem Cell Therapy An Option?
As previously mentioned, there is no curable treatment for ALS available. However, scientists are researching Stem Cell Therapy as the new favorable approach in the treatment of neurologic disorders.
There is a rising interest in Stem Cell Therapy as a promising remedy for curing ALS. Mesenchymal stem cells are particularly believed to be the most suitable ones due to their availability, absence of ethical issues and positive results in various experiments.
Studies and clinical trials have begun to apprehend the benefits of MSC transplantation. They demonstrate that MSCs lead to a partial recovery of motor neurons and a delay in disease progression. Also, there has been no evidence of a major adverse effect after MSC transplantation.
When testing this newfound research on animals, the lifespan of the subjected animal has increased with MSC transplantation. These positive results have encouraged the administration of MSC in ALS patients.
However, despite the safe outcomes of MSC transplantation in humans, results show that there is only a partial improvement in ALS sufferers with only a few cases that showed a delay in disease progression. Hence, there is a need for further studies and trials on a higher number of human subjects for a better understanding of MSC effects so that more significant conclusions can be reached.
- Published in Blog
The Keto Diet: A Miracle Formula Or A Dangerous Risk?
We live in a world overloaded with diets and eating plans that are all preaching miracle, long term results for weight loss and reducing risks of developing illnesses and diseases.
But, with so many different plans out there all claiming to produce the best results, it’s hard to know which one to recommend to your patients. The Paleo, the Atkins, the Pritikin Principle, the South Beach, WeightWatchers, The Zone Diet… the list goes on and on. Recently, many doctors are leaning more towards THE KETO DIET as a recommendation for not only their overweight patients; but patients that suffer from various illnesses and ailments.
WHAT IS A KETO DIET?
To put it simply, a ‘keto’ or ‘ketogenic’ diet is a low-carb, high-fat diet.
A reduction in carbohydrates puts the body into a metabolic state called ketosis. This is when the body produces small fuel molecules called ketones. Carbs are broken down into glucose and used for energy. So when someone deprives themselves of carbs, the body has no choice but to switch its fuel supply from glucose (blood sugar) to ketones found in body fat.
Therefore, as long as carbs are restricted, the body is constantly burning fat.
Kathleen M. Zelman, MPH, RD, LD told WebMD that a ketogenic diet is often used for losing weight, but it can help manage certain medical conditions such as epilepsy, heart disease, diabetes type 2, brain diseases and IBS.
WHAT ARE THE MAIN BENEFITS OF A KETO DIET?
Weight Loss & Appetite Suppression
The biggest benefit of the keto diet and the one most dieters are striving for is weight loss and suppression of appetite.
Typically, the body takes their fuel from our stores of glycogen, but with the reduction of carbohydrates in a keto diet, glucose levels are lowered and the body is forced to obtain its energy from body fat. This results in patients experiencing weight loss.
Food cravings and hunger is often the reason that our patients are failing at dieting. Another key reason why medical professionals have found the keto diet to be so successful is because of the effects it has on reducing cravings and tackling hunger.
Studies have proven that participants that follow a low-carb diet have reported a decrease in appetite and cravings compared to individuals following a low-fat diet. This is possibly down to high-fat foods slowing down the process of gastric emptying. Food that is high in fat passes through the gut at a much slower pace, leaviing the individual feeling satisfied for longer.
Diabetes Type 2
Diabetes Type 2 is triggered by spikes occurring in the patient’s blood sugar levels. Insulin is used to regulate and control these spikes. When a patient with type 2 diabetes follows a keto diet, the amount of glucose they are consuming dramatically decreases due to the lower intake of carbohydrates.
So the lower intake of glucose found in high carb food = the lower intake of insulin.
A study of 21 overweight men with type 2 diabetes who were put on a keto diet showed that most participants saw a reduction in their insulin medication or were taken off diabetes medication completely as a result of controlling their glucose levels by following a keto diet.
Epilepsy
A keto diet has been used to treat epilepsy since the 1920s and during a recent study, seventy percent of epileptic patients have reported less and more controlled seizures and a reduction in their medication since implementing a keto diet.
Epileptic seizures occur when a network of neurons fire unexpectedly due to the overexcitement of brain cells releasing neurotransmitters, like glutamate. The brain is then not able to suppress the spread of excitability like in non-epileptic people using inhibitory neurotransmitters like gamma-aminobutyric acid, or GABA.
What a keto diet can do to control seizures is to reduce the amount of glutamate in the brain and enhance the synthesis of GABA.
Alzheimer’s Disease & Dementia
A ketogenic diet can contribute towards keeping the brain young and healthy and reduce the risk of developing degenerative brain diseases such as Alzheimer’s disease and dementia.
Findings from a study found that keto diets can protect neurovascular function against cognitive decline by clearing out beta-amyloid protein in the brain. Beta-amyloid creates toxic plaques that interfere with neuronal signaling that results in Alzheimer’s disease and dementia. Improvements to blood flow to the brain, which contributes to healthy cognitive function, were also noted.
IBS & Other Gastronomical Conditions
Fifteen percent of the general population claim to suffer from IBS. It is one of the most common complaints taken to doctors. A lot of research has been undertaken in recent years around the effect that carbohydrates have on the gastrointestinal system.
Studies have found that when sufferers consume carbohydrates, they tend to ferment in the small intestine and causes gas and bloating. For IBS sufferers, carbs are poorly absorbed by the gut wall, causing fluid to remain in the intestinal space, resulting in diarrhea.
Therefore, when IBS sufferers follow a keto diet and reduce their intake of carbohydrates, they report significant improvements to their condition.
HOWEVER…
All these amazing results from consuming low-carb, high-fat foods sounds promising, right?
Well…
Medical professions at Harvard Medical School are urging medical professionals to be wary of recommending the keto diet to their patients. They have identified numerous risks associated with following a ketogenic diet.
Not only is the body being deprived of an essential food group – carbohydrates, but a keto diet is typically high in saturated fat. High levels of saturated fat can lead to high cholesterol and heart disease.
Other risks they have been identified with following a keto diet is:
A deficiency in micronutrients including selenium, magnesium, and vitamins B and C which are found in most of the fruit and vegetables that are restricted in a keto diet.
Liver problems due to the amount of fat the body needs to metabolize. This can cause existing liver problems to worsen.
Kidney problems due to the amount of protein the body needs to metabolize. A keto diet can put strain on the kidneys and make exisitng conditions worse.
Constipation. The keto diet is low in fibrous foods like grains and legumes. The digestive system relies on a steady flow of fiber to break down foods and get rid of body waste.
“The Keto Flu” is a term for the mood swings, body aches and brain fuzz that people experience when following a keto diet. The brain needs glucose to function and when it is deprived of this, it can cause irritability and tiredness.
We want to know your thoughts and opinions on the Keto Diet!
Have you ever recommended the keto diet to your patients? Do you see the benefits happening in your patients? Is it dangerous?
- Published in Blog
James P. Allison, PhD. Nobel Prize in Medicine
JAMES P. ALLISON, PhD
Chair, department of immunology, and director.
Immunotherapy Platform, MD Anderson Cancer Center
Losing his mother to lymphoma and two uncles to melanoma and lung cancer taught James P. Alli- son, PhD, a difficult lesson at an early age. “My mother was treated with radiation and my uncle, who had lung cancer, was treated with chemotherapy. I saw the ravages of those treatments, which ultimately were unsuccessful,” he says.
Allison knew he wanted to work in cancer research, developing more effective and less toxic therapies. An undergraduate course at the University of Texas at Austin sparked his interest in T cells—warrior cells of the immune system that defend the body against infections and cancer.
The next step, he says, was to pre vent the brakes from engaging—“to give the T cells time to keep going and eliminate the tumors.” Allison’s work led to the development of drugs called checkpoint blockade antibodies, including ipilimumab (Yervoy) for melanoma. “When we started this work, the median life expectancy with metastatic melanoma was 11 months, and no drug had ever changed that,” he says. Among people treated with Yervoy, more than 20% are still alive three years later, and some have survived 10 years.
New drugs targeting another off-switch, PD-1, have since been developed to treat cancers of the head and neck, lung, kidney, and bladder, among others. Now Allison’s lab is studying different combinations of checkpoint blockades to see which patients will respond best to them. “I don’t think these approaches are going to replace any. of the traditional therapies,” he says, but, “I think that soon, immunotherapy is going to be part of every successful cancer therapy.”
- Published in Blog
Opioid Overdose Crisis in America.
Let’s face it; it’s like America is missing it by paying more attention to Gun Violence murder and other deadliest diseases than opioid addiction that leads to an average number of deaths of 130 people every day. There is no doubt; the United States is in the midst of an opioid crisis, as overdose and addiction of opioids turns out to be a serious national crisis that has reduced the life expectancy of the US. With an average victim’s death hitting over 47,000 people recorded in 2017.
However, despite the alarming rate of death from opioid overdose, this problem is not grabbing the headlines from the media for health stakeholders to pay more attention and look for ways to finding lasting solutions to the anomaly.
It is worthy to note that, while many people are addicted to opioids in the form of heroin and synthetic opioids, millions of people are out there, taking an overdose of opioids in the form of pain reliever prescription for chronic pains and other related health issues. Though, opioids are used in the treatment of chronic pain, research has suggested that they may not help relieve pain in the long-term. This has posed a serious threat to public health and economic welfare, not only for the United States but other affected countries as well.
According to the Centers for Disease Control and Prevention, the total “economic burden” of only opioid misuse prescription in the US is estimated as $78.5 billion per year, inclusive of lost productivity, criminal justice involvement, healthcare cost, and addiction treatment.
This article aims to look at how we come to this point, some facts about opioids in the United States, and some useful information about opioid overdose. Read on to uncover them all.
Opioids and Substance Abuse at a Glance.
Opiates, popularly known as narcotics, are commonly prescribed for quick pain relief and sleep inducement. Originally, it is derived from poppy plant seeds or their byproducts. Opiates occur naturally in the form of opium and morphine, but most opiates are synthetic. These drugs became highly addictive because they create an intense sense of euphoria and as well as safeness, when adding it to pain-relieving properties.
Most patients with pain disorders later turn to rely on pharmaceutical opiates like oxycodone and hydrocodone; hence, they become addicted to it. However, opioid overdose has been studied to cause a number of health problems in the users, and these health issues are not limited to serious disorders, but even death. One of the prolonged effects of opioid usages is the brain’s inability to produce endorphins naturally. Endorphins are known to be the body’s natural painkillers.
Initially, around late 1990s, when opioids were becoming popular among the people, pharmaceutical companies came out to allay the fear of the people and reassure the medical community that opiate users would not become addicted to opioids prescribed for pain relief; hence, healthcare service providers started prescribing the drugs to patients at greater rates. Subsequently, this act resulted in a extensive misuse and diversion of these drugs prior to the time it was discovered that opioid prescriptions could actually be highly addictive like other opiates.
Facts about Opioid Prescription and Misuse in America.
The rates of opioid overdose in the United States started increasing in 2017 with over 47,000 citizens died due to misuse of opioids, such as heroin, prescription opioids, illicitly manufactured fentanyl (one of the powerful opioid synthetics). In the same year, this number was estimated 1.7 million and 652,000 Americans suffered from prescription opioid pain relievers and heroin use disorder respectively. The question is, how does it start?
It all starts when the body cannot properly manage and regulate pain again. In this condition, an opiate user may become addicted to the drugs, as the drugs now used to relief their pain and at the same time, create a sense of contentment and happiness in using them. Over time, even after the pains have gone, an opiate user will require more of the substance to reach the same level of high that creates happiness and contentment which they first experienced; hence, the person is already “an opiate addict.” Withdrawing from its use makes it even worse, as their bodies begin to show some unpleasant symptoms that make the user seeking more to relieve the symptoms.
From here, opioid prescription and misuse have turned out to become a public health problem in the United States with alarming rates of death on a daily basis.
Below are some facts about the opioid crisis in the United States:
- According to WQAD Digital Team’s claim of IMS Health’s market research, the number of opioid prescriptions doctors dispensed increased from 112 million to 282 million from 1992 to 2012 respectively. However, according to IQVIA, the number has declined to 236 million in 2016 and further dropped by 10.2% in 2017.
- Centers for Disease Control and Prevention, about 68% of cases of estimated 70,200 drug overdose deaths recorded in 2017 were linked to the use of opioids. This is more than six times compared to that of 1999 (including illegal opioids such as heroin and illicitly manufactured fentanyl and prescription opioids).
- According to a review, about 21 – 29% of patients with chronic pain who take opioids prescription misuse them.
- Another study also claimed that roughly 80% of heroin users initially misused prescription opioids.
- The same study also claimed that about 8 – 12% of opiate users develop an opioid use disorder.
- According to the Centers for Disease Control and Prevention, there is a 30% increase in opioid overdoses in 45 states of the United States between July 2016 to September 2017.
- In another review, opioid overdoses were seen increasing by 70% in the Midwestern region between July 2016 to September 2017.
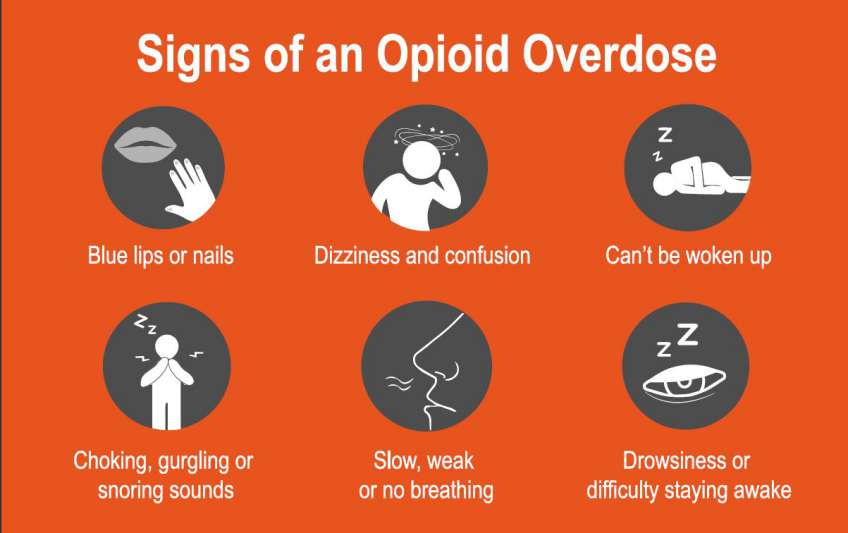
What is been doing about it?
With over two million opioid dependants in the United States, the U.S. Department of Health and Human Services (HHS) is making efforts to tackle the opioid crisis in America by focusing on five major areas. These include:
- Promoting and enlighten people on the use of overdose-reversing drugs.
- Giving people access to treatment and recovery therapies.
- Offering support for modern research on addiction and pain.
- Promoting public health surveillance to help people understand the epidemic better.
- Enhancing better pain management practices in the country.
In furtherance to the efforts of the U.S. Department of Health and Human Services (HHS), the National Institutes of Health (NIH)’s Director Francis S. Collins in April 2018’s National Rx Drug Abuse and Heroin Summit, announced the launch of a special program referred to HEAL (Helping to End Addiction Long-term) Initiative. HEAL is an aggressive effort set up to expedite scientific solutions to address the opioid crisis in the United States.
Final Words.
Just because media are not paying attention to the opioid crisis in the US does not mean things are working in the medical world. With the alarming rate of opioid overdose and an increasing number of Americans abusing prescription and becoming dependent on opioids, all hands must be on deck to find a lasting solution to the current problem. While different agencies and stakeholders are not relenting on their efforts, more medical developments and approaches are still required to achieve a good result.
- Published in Blog
Autologous mesenchymal stem cell application for cartilage defect in recurrent patellar dislocation: A case report
Introduction.
Recurrent patellar dislocation is a repeated dislocation that follows from an initial episode of minor trauma dislocation . Conservative management gives a minimal result in re-dislocation, with persistent symptoms of anterior knee pain, instability and activity limitation. Meanwhile, there is no gold standard treatment of realignment procedures. This can further cause cartilage lesion in the patella and femoral condyle, and consequently increase the risk of re-dislocation. Mesenchymal stem cells (MSCs) have been widely explored for treating cartilage defect due to their potency of chondrogenic differentiation. We present a novel approach of treating cartilage lesions in recurrent patellar dislocation by combining of arthroscopic microfracture and autologous bone marrow derived MSCs (BM-MSCs) after Fulkerson osteotomy.
Presentation of case.
A 21-year-old male presented with left knee discomfort. Ten years ago, the patient felt discomfort on the medial side of the knee and felt his knee cap slide out laterally. The patient experienced several episodes of instability ranging from a feeling of “giving away” until a prominent lateral sliding-off of his knee cap. Anterior knee pain has also occurred during activities such as climbing stairs or exercising.
Physical examination revealed slight pain on the anterior side of the patella, but no atrophy or squinting patella. Knee range of motion (ROM) was normal when the knee cap position was normal, but was limited when it was dislocated (0–20°). Lateral subluxation of the patella was found when the knee was extended from 90° flexion position (J-sign positive), positive patellar apprehension test, with medial patella elasticity/patellar glide >2 quadrants. The Q angle, in the 90° flexed knee position, was 10°, which was still normal. The plain radiograph imaging showed no abnormality. Insall-Salvati index was 1.12. The patient was diagnosed with recurrent patellar dislocation, with suspected cartilage lesion of the left knee.
The first surgery was an arthroscopy diagnostic and distal realignment procedure (lateral retinaculum release, percutaneous medial retinaculum plication, and antero-medialization of tibia tubercle/Fulkerson osteotomy). We found articular cartilagedefects on the lateral condyle of the femur with a diameter of 3 cm (Figure. 1A), and on the postero-medial with a diameter of 2.5 cm (Figure. 1B), and the depth of both was more than 50% of the cartilage thickness. We determined that the articular defect was Grade 3 according to International Cartilage Regeneration & Joint Preservation Society (ICRS). We performed a dissection of lateral retinaculum (lateral release) (Figure. 1C) using an electrocautery, continued by incising the medial side of tibia tuberosity and detaching the patellar tendon by using an oblique osteotomy procedure on tibia tuberosity, where the fragment slide 1 cm antero-medially and fixed with two 3.5 mm (length 40 mm) partial threaded cancellous screw, followed by percutaneous plication on the medial side of the patella using non-absorbable string (Figure. 2A). Post-operative ROM was 90° flexion without any dislocation (Figure. 2B) and the position of the screws was good (Figure. 2C).

Figure 1.A. Cartilage defect on the femoral lateral condyle with a diameter of 3 cm (pointed by the arrow). B. Articular cartilage defect on posteromedial patella with a diameter of 2.5 cm (pointed by the arrow). C. Lateral retinaculum dissection/lateral release using an electrocautery (pointed by the arrow).

Figure 2.A. Percutaenous medial plication using non-absorbable string no.2. B. Post-operative anteroposterior and lateral projection of plain radiograph imaging. C. Post-operative CT scan.
One month after surgery, full ROM and weight bearing exercises were started, including knee exercise until maximum flexion was reached along with quadriceps muscle exercise. Eighteen month after that surgery, we performed an iliac crestbone marrow aspiration; arthroscopic microfracture by using an awl until 4 mm depth was reached on the site located ±3–4 mm from the articular cartilage defect on the posteromedial patella and femoral lateral condyle (Fig. 3A); and tibial tuberosity screw removal.

Figure 3.A. Arthroscopic microfracture on cartilage defect using an awl, with a depth of ±4 cm. B. The mesenchymal stem cell culture after day 22 showing fibroblast-like cell/spindle shaped cells that 100% confluent.Approximately 30 mL of bone marrow was aspirated from the posterior iliac crest. Bone marrow aspirate was diluted in phosphate-buffered saline (PBS) and centrifuged at room temperature. The buffy coat was washed and cultivated for 3–4 weeks until reaching the required amount (107 MSCs/mL) (Figure. 3B). The cells were harvested and characterized with flow cytometer. The MSCs, having negative bacteria and fungi tests, were injected intra-articularly into the left knee. Then, 2 mL HA were injected weekly for 3 weeks. Non-weight bearing exercise was conducted for 6 weeks.
Outcomes were assessed by using International Knee Documentation Committee (IKDC) score, visual analog scale (VAS) score and imaging. Baseline IKDC score was 52.9 and VAS score was 8. Nineteen months after the first surgery, IKDC score was improved to 93.1, while the VAS score decreased to 2. Six months after MSCs implantation, evaluation by MRI FSE cor T2-weighted signal (cartilage sequence) showed a significant growth of articular cartilage covering most of the defect (Figure. 4). Two years after the MSCs implantation, there was no complaint and full ROM was reached.

Figure 4.MRI FSE cor T2-weighted signal in different slice. Left image showing cartilage defect (pointed by the arrow). Right image showing cartilage growth was found in the defect (pointed by the arrow).
Discussion
Recurrent patellar dislocation are uncommon problem, with recurrence rate 15%–44% after conservative management, while cartilage lesions following recurrent patellar dislocations are quite common, but still no gold standard or consensus on the management. This patient was diagnosed as chondromalacia Grade 3 Outerbridge classification and Grade 3 ICRS. One of the suitable procedures for recurrent patellar dislocation with chondromalacia, especially Grade 3 or 4, was Oblique Fulkerson-type osteotomy, with or without the release of lateral retinaculum. This distal realignment procedure could decrease patellofemoral pain by anteriorization of tibial tuberosity, decreasing articular contact pressure and at the same time medializing knee extensor mechanism. Therefore, we performed the Fulkerson-type osteotomy with lateral retinacular release, combined with percutaneous medial plication since the patient was already 21 years of age and the bone was expected to be mature so that the risk of premature physeal closure in proximal tibia can be avoided. This technique has demonstrated good results (86%), although it had a risk of tibial stress fracture in the healing process. The lateral retinacular release is an adjuvant after tibial tubercle medialization to re-center the patella. It was reported that isolated lateral retinacular release significantly gives an inferior long-term result compared to medial reefing. Percutaneous plication of medial patella procedure was indicated to build a strong construct by shortening the patellofemoral ligament, in order to prevent lateral sliding of the patella.
Treatment of articular cartilage defect remains challenging since it has limited self-healing capacity. Lesions that do not reach the subchondral zone will be unlikely to heal and usually progress to a cartilage degeneration. Limited blood supply in the cartilage and low chondrocyte metabolic activity disrupt natural healing that is supposed to fill the defect by increasing hyaline cartilage synthesis activity or stem cell mobilization from bone marrow to site of injury. The proper initial procedure for chondral lesion >4 cm2 was marrow stimulation by mosaicplasty or microfracture; and for a lesion <4 cm2 and >12 cm2 accompanied with symptoms, autologous cartilage implantation (ACI) beneath a sutured periosteal flap was promising. This procedure could not regenerate cartilage in the long term, due to loss of flap or cell suspensions. A scaffold (e.g. HA) was then used to act as an anchorage for chondrocytes adherence on cartilage defects and to promote the secretion of chondrocyte extracellular matrix. The BM-MSCs implantation could be an alternative source of the chondrocytes. Human BM-MSCs are relatively easy to isolate and to be cultured in such a condition that may retain their capability to differentiate into chondrocytes.
The MSCs effect was reported as effective as ACI and even had the advantage over ACI in terms of the number cells obtained, better proliferation capacity and less damage in the donor site. Treating large cartilage defects by using BM-MSCs showed good outcome, but the transplantation procedure was invasive. Wong et al. conducted a clinical study of the BM-MSCs intra-articular injection in combination with high tibial osteotomy (HTO) and microfracture for treating cartilage defect with varus knee. They reported that intra-articular MSCs injection improved the outcomes in the patients undergoing HTO and microfracture. Here we performed also a less invasive approach by injecting the autologous BM-MSCs intra-articularly, following the arthroscopic microfracture using an awl to penetrate the subchondral bone plate in the cartilage defects, which led to clot formation. This clot contains progenitor cells, cytokines, growth factors and pluripotent, marrow-derived mesenchymal stem cells, which produce a fibrocartilage repair with varying amounts of type-II collagen content. Cytokine within the fibrin clot will attract the injectable stem cells to the cartilage lesions.
The HA injection in this patient was aimed to suspend the MSCs and to support regenerative potency of MSCs with chondroinductive and chondroprotective potency of HA. Intraarticular injection of MSCs suspended in HA could be an alternative treatment for large cartilage defect. Supporting microfracture technique by intra-articular HA injections had a positive effect on the repair tissue formation within the chondral defect. The MRI showed that there was a growth of articular cartilage covering most of the defect even though it was not perfect as yet.
Conclusion
This case report demonstrated that combining Fulkerson osteotomy with the lateral retinacular release and percutaneous medial plication was effective in treating chronic patellar instability. The combination of microfracture and MSCs implantation was safe and could regenerate the articular cartilage in this patient.
- Published in Blog
Autologous micro-fragmented adipose tissue for the treatment of diffuse degenerative knee osteoarthritis: an update at 3 year follow-up
Background.
The management of chondral disease is challenging because of its intrinsic poor healing potential. Biomechanical and biological changes may lead to the loss of tissue homoeostasis, resulting in an accelerated degeneration of the articular surface, eventually leading to end-stage osteoarthritis (OA).
Conservative therapies for the treatment of knee degenerative processes, such as non-pharmacological interventions, systemic drug treatment and intra-articular therapies are used before resorting to surgery; nonetheless, they may offer only short-term benefits. Encouraging preliminary results have been reported using mesenchymal stem cells (MSCs), either alone or in association with surgery. Among the many sources of MSCs, adipose tissue has created a huge interest in the context of cartilage regeneration (Pak et al. 2016; Ruetze and Richter 2014), due to its wide availability, ease to harvest and richness in mesenchymal cell elements within the so called stromal vascular fraction (De Girolamo et al. 2016; Caplan 2008; Caplan and Correa 2011; Caplan and Dennis 2006). Moreover, MSCs from adipose tissue are characterized by marked anti-inflammatory and regenerative properties, which make them an excellent tool for regenerative medicine purposes (De Girolamo et al. 2016; Caplan 2008; Caplan and Correa 2011; Caplan and Dennis 2006). Nevertheless, preparation of autologous MSCs for injection requires ex vivo culture from a good manufacturing practice facility, which makes the process laborious and expensive (Ährlund-Richter et al. 2009; Arcidiacono et al. 2012; Sensebé et al. 2010). An increasing number of adipose tissue-derived cell isolation systems, allowing for minimal manipulation, have been developed in the last years. We previously reported the safety and feasibility of autologous micro-fragmented adipose tissue as adjuvant for the surgical treatment of diffuse degenerative chondral lesions at 1 year follow-up (Russo et al. 2017). Here we present the outcomes of the same cohort of patients evaluated at 3 year follow-up.
Methods.
The original study was approved by the Ethics Committee of Verona and Rovigo – Italy (protocol n° 10,227, March 1st, 2016). An extension of the study protocol has been conceded by the same authority to evaluate the results at 3 years (protocol n° 14,505, March 14th 2018) and written informed consent was obtained from all patients.
Study design and population, surgical techniques, post-op rehabilitation protocol, safety and clinical evaluation were previously described (Russo et al. 2017). Briefly, 30 patients, affected by diffuse degenerative chondral lesions of different degrees of severity, were treated with autologous and micro-fragmented adipose tissue between 1stJanuary 2014 and 31st December 2014. Of these 30 patients, 24 (80%) also had an associated surgery (ACL/LCL reconstruction, high tibial osteotomy, meniscectomy), while six (20%) underwent arthroscopy alone. For the 3 year follow-up all the patients were re-contacted and clinically evaluated by the same clinicians.
Findings.
Of the 30 patients treated with autologous micro-fragmented adipose tissue, eight also had meniscal surgery, five plate removal, three osteotomy, two ligament surgery, two microfractures, and four other surgical procedures. The remaining six had arthroscopy alone. Despite the heterogeneity of the associated surgical procedures all the patients shared the presence of chondral lesions of different degrees of severity (Russo et al. 2017).At 3 years follow-up, one patient was lost, and seven (23%) received additional treatments in the period of observation, and therefore have been considered failures. In detail, between 18 and 30 months, one patient had three injections of hyaluronic acid and the other six had multiple injections of platelet rich plasma. Background data on this subpopulation is reported in Table 1.
Table 1: Background data of the failures (n = 7)
| Age y/o | |
| Mean | 36.3 |
| Standard deviation | 7.3. |
| Type chondropathy | |
| FC | 4 (57%) |
| TP | 2 (29%) |
| PF | 6 (86%) |
| Three-compartment | 2 (29%) |
| Associated surgery | |
| YES | 5 (71%) |
| NO | 2 (29%) |
FC femoral condyle, TP tibial plateau, PF patellofemoral.
No adverse events, lipodystrophy cases at the harvesting site nor atypical inflammatory reactions at the joint level were reported in the 3 year period for all the 29 patients.On average, the 22 patients that had no other treatments in the 3 year period (Table 2) showed that the results observed at 1 year were maintained (T36 vs. T12, p > 0.05). Moreover, 41, 55, 55 and 64% of the patients improved with respect to the 1-year follow-up in the Tegner Lysholm Knee, VAS, IKDC-subjective and total KOOS, respectively.
Table 2: Background data of the population (n = 22)
| Age y/o | |
| Mean | 44.7 |
| Standard deviation | 11.4 |
| Gender | |
| M | 14 (64%) |
| F | 8 (36%) |
| BMI | |
| Mean | 25.9 |
| SD | 3.3 |
| Sport | |
| Profesionals | 1 (4%) |
| Amateurs | 9 (41%) |
| Occasional | 5 (23%) |
| Inactive | 7 (32%) |
| Grade chondropathy (ICRS classification) | |
| II | 7 (32%) |
| III | 6 (27%) |
| IV | 9 (41%) |
| Type chondropathy | |
| FC | 17 (77%) |
| TP | 14 (64%) |
| PF | 14 (64%) |
| Three-compartment | 9 (41%) |
| Associated surgery | |
| Yes | 18 (82%) |
| No | 4 (18%) |
FC femoral condyle, TP tibial plateau, PF patellofemoral. Compared to pre-operative values, more than 50% of the patients improved at least 20 points in all the considered scores, and, surprisingly, 55% of the patients improved at least 30 points in the VAS pain scale.
A summary of the results is reported in Figure 1.

Figure 1: Trend of functional improvements of Tegner Lysholm knee, VAS pain, IKDC subjective and total KOOS pre-operatively (white bars), at 12 (grey bars) and 36 months (black bars) after micro-fragmented adipose tissue injection. Results are expressed as mean and standard error.
Discussion
The main finding of this study is that the beneficial effect of autologous micro-fragmented adipose tissue as adjuvant for the treatment of diffuse degenerative chondral lesions is maintained in the mid-term. In addition, no complications were observed in the 3 year period showing the safety profile of this procedure. No patient, including the seven patients who received additional treatments, worsened compared to the pre-operative condition.
Despite the heterogeneity of the associated surgical procedures all the patients shared the presence of chondral lesions of different degrees of severity, which may have been responsible for the impairment in function and pain.
As reported in literature, articular surface damages, especially when diffused (three compartment OA), positively correlate with a decay in the outcomes in patients who received knee surgery for other reasons (Bonasia et al. 2014; Røtterud et al. 2012; Saithna et al. 2014; Su et al. 2018; Verdonk et al. 2016). Published data shows a decline in the clinical results in the mid to long-term for arthroscopic and chondral debridement procedures in cases of initial knee OA (Su et al. 2018). Some authors assessed the effectiveness of the arthroscopic or conservative treatments in patients diagnosed with knee OA (Kellgren-Lawrence grade 2 to 4) with 5 years of follow-up, concluding that arthroscopy provided no benefit in decreasing or delaying arthroplasty and that it can relieve symptoms only up to 2 years (Su et al. 2018). The same observation has been reported for ligament reconstruction, where the short and mid to long-term benefits are inferior in patients who have cartilage lesions. In a study of a cohort of ACL-injured patients with full-thickness cartilage lesions (ICRS grade III–IV), the authors showed that ACL-injured patients with full-thickness cartilage lesions reported worse outcomes and minor improvement after ACL reconstruction compared to patients without cartilage lesions at 2–5 years follow-up, although no significant differences between the two groups at the time of ACL reconstruction were present. This means that the observed differences between the groups must have occurred during the follow-up period (Røtterud et al. 2012). Furthermore, the outcomes of osteotomy procedures in patients with diffuse degenerative knee chondropathy worsen in the mid to long-term (Bonasia et al. 2014; Saithna et al. 2014). In a study reporting the results of a case series of opening wedge distal femoral varus osteotomies for valgus lateral knee OA, it is shown that re-operation for non-arthroplasty related surgery was common due, besides others, to infection and persistence of symptoms (Saithna et al. 2014). With regard to meniscectomy, in a recently published paper it is concluded that meniscus therapy including partial meniscectomy, meniscus suture, and meniscus replacement has proven beneficial effects in long-term studies in patients without cartilage damage, supporting the hypothesis that meniscectomy increases the risk of cartilage degeneration (Verdonk et al. 2016).
Based on the aforementioned published evidences, we should have expected, in the mid-term, a decay of the outcomes. Notably, the results have been maintained with no significant differences in all the evaluated parameters with respect to the 1 year follow-up assessment. Furthermore, in line with that already observed at 1 year, the patients with lesions in more than one compartment had higher and statistically significant improvements compared to patients with lesions in only one compartment (p < 0.01). This finding supports our hypothesis of using micro-fragmented adipose tissue for the treatment of the diffuse degenerative knee pathology as an adjuvant of the surgical procedures. Indeed, the maintenance of stable results at the last follow-up leads to hypothesize a protective role of micro-fragmented adipose tissue in a further chondral degeneration.
The seven patients who received additional biological therapies in the 3-year period, were young (mean age 36.3 ± 7.3 vs. 44.7 ± 11.4), very active in sport and 6 out of 7 had a patellofemoral chondropathy. Their conditions after 1 year did not worsen, but they probably needed an additional biological treatment because of their high functional demands and the presence of the patellofemoral chondropathy, which is a negative prognostic element, even if the small number of patients does not allow for any statistical correlation.
References.
- Ährlund-Richter L, De Luca M, Marshak DR, Munsie M, Veiga A, Rao M (2009) Isolation and production of cells suitable for human therapy: challenges ahead. Cell Stem Cell 4(1):20–26View ArticleGoogle Scholar
- Arcidiacono JA, Blair JW, Benton KA (2012) US Food and Drug Administration international collaborations for cellular therapy product regulation. Stem Cell Res Ther 3(5):1View ArticleGoogle Scholar
- Bonasia DE, Dettoni F, Sito G, Blonna D, Marmotti A, Bruzzone M, Castoldi F, Rossi R (2014) Medial opening wedge high tibial osteotomy for medial compartment overload/arthritis in the varus knee: prognostic factors. Am J Sports Med 42(3):690–698View ArticleGoogle Scholar
- Caplan AI (2008) All MSCs are pericytes? Cell Stem Cell 3(3):229–230View ArticleGoogle Scholar
- Caplan AI, Correa D (2011) The MSC: an injury drugstore. Cell Stem Cell 9(1):11–15View ArticleGoogle Scholar
- Caplan AI, Dennis JE (2006) Mesenchymal stem cells as trophic mediators. J Cell Biochem 98(5):1076–1084View ArticleGoogle Scholar
- De Girolamo L, Kon E, Filardo G, Marmotti A, Soler F, Peretti G, Vannini F, Madry H, Chubinskaya S (2016) Regenerative approaches for the treatment of early OA. Knee Surg Sports Traumatol Arthrosc 24(6):1826–1835View ArticleGoogle Scholar
- Pak J, Lee JH, Kartolo WA, Lee SH (2016) Cartilage regeneration in human with adipose tissue-derived stem cells: current status in clinical implications. Biomed Res Int 2016:4702674View ArticleGoogle Scholar
- Røtterud JH, Risberg MA, Engebretsen L, Årøen A (2012) Patients with focal full-thickness cartilage lesions benefit less from ACL reconstruction at 2–5 years follow-up. Knee Surg Sports Traumatol Arthrosc 20(8):1533–1539View ArticleGoogle Scholar
- Ruetze M, Richter W (2014) Adipose-derived stromal cells for osteoarticular repair: trophic function versus stem cell activity. Expert Rev Mol Med 16:e9View ArticleGoogle Scholar
- Russo A, Condello V, Madonna V, Guerriero M, Zorzi C (2017) Autologous and micro-fragmented adipose tissue for the treatment of diffuse degenerative knee osteoarthritis. J Exp Orthop 4(1):33View ArticleGoogle Scholar
- Saithna A, Kundra R, Getgood A, Spalding T (2014) Opening wedge distal femoral varus osteotomy for lateral compartment osteoarthritis in the valgus knee. Knee 21(1):172–175View ArticleGoogle Scholar
- Sensebé L, Bourin P, Tarte K (2010) Good manufacturing practices production of mesenchymal stem/stromal cells. Hum Gene Ther 22(1):19–26View ArticleGoogle Scholar
- Su X, Li C, Liao W, Liu J, Zhang H, Li J, Li Z (2018) Comparison of arthroscopic and conservative treatments for knee osteoarthritis: a 5-year retrospective comparative study. Arthroscopy 34(3):652–659View ArticleGoogle Scholar
- Verdonk R, Madry H, Shabshin N, Dirisamer F, Peretti GM, Pujol N, Spalding T, Verdonk P, Seil R, Condello V (2016) The role of meniscal tissue in joint protection in early osteoarthritis. Knee Surg Sports Traumatol Arthrosc 24(6):1763–1774View ArticleGoogle Scholar
- Published in Blog