Global Stem Cells Group announces the opening of a new Stem Cell Center in Bandung, Indonesia
Miami, FL, December 12, 2022 – Global Stem Cells Group (GSCG) is pleased to announce the opening of a new Stem Cell Center in Bandung, Indonesia in partnership with the Dr. Yanti Aesthetic Clinic. This joint venture will be the second in the country as they already have another facility in the stunning City of Surabaya.
The New Global Stem Cell Group Facility is Expected to Transform the Regenerative Medicine Field
The GSCG’s new center in Bandung aims to increase awareness about the benefits of regenerative medicine. Its launch strengthened the relationship between GSCG and the Dr. Yanti Aesthetic Clinic. This new facility is expected to transform regenerative medicine in the following ways:
- Offering accessible stem cell therapy to Indonesians;
- Promoting regenerative medicine technology; and
- Inspiring other doctors and industry experts to explore regenerative medicine.
Top Government Officials Attended the Launch of the Global Stem Cell Group Office in Bandung
The launch of the new Global Stem Cell Group facility in Bandung was a remarkable event in the medical field. The Minister of Tourism was among the top officials that witnessed the launch. Furthermore, the office of the Vice President sent several representatives.
What the President of Global Stem Cell Group Has to Say
Benito Novas, the president of the GSCG, was among the top industry specialists that graced the launch of the company’s second facility in Indonesia. According to him, GSCG wants to make regenerative medicine readily available for patients worldwide. Likewise, the company is encouraging more doctors to adopt stem cell therapy’s clinical and aesthetic applications in their work.
Benito Novas said: “We are dedicated to making it possible for both doctors and patients in all parts of the world to experience the benefits of regenerative medicines. The company is expanding and establishing itself as a market leader.”
The Director of the Dr. Yanti Aesthetic Clinic Appreciates GSCG
Dr. Yanti Kushmiran, director of the DR. Yanti Aesthetic Clinic, is once again honored to be part of this step into the future of regenerative medicine with its second Stem Cell Center in Indonesia together with the leading company Global Stem Cells Group.
Regarding this new clinic, Dr. Yanti says “We are honored to be part of GSCG, which has a more than 10-year track record in the market and a strong international reputation and to open a second cell therapy and regenerative medicine center facility under the Stem Cell Center brand. We will be able to provide more services to our patients as a result of this new partnership.”
Another GSCG Clinic is Opening in Indonesia by January 2023
The Global Stem Cell Group will continue increasing patients’ access to advanced regenerative medicine. As a result, the company plans to open another branch in Jakarta, the Capital of Indonesia. GSCG and its partners plan to launch this third facility in January 2023.
About Global Stem Cell Group
The Global Stem Cell Group is a family of several companies focused on stem cell medicine and research. The company uses its network to bring leadership in regenerative medicine training, research, and patient applications.
GSCG’s mission is to allow physicians to present the benefits of stem cell medicine to patients worldwide. The company also partners with policymakers, educators, and regulators to promote regenerative medicine.
Global Stem Cells Group is a publicly traded company operating under the symbol MSSV. https://finance.yahoo.com/quote/mssv/
To learn more about Global Stem Cells Group, Inc.’s companies visit our website www.stemcellsgroup.com or call +1 305 560 5331
- Published in News
Global Stem Cells Group, Announces Launch of New Stem Cells and Regenerative Medicine Clinic in CDMX
Miami, FL, October 28, 2022 – Global Stem Cells Group announces a new partnership that enhances its goal of establishing its therapies and technology to meet market demand in populated areas of the world.
This collaboration with STEM LIFE clinic’s new facility and Dr. Vanessa Rodriguez Pares, currently one of the most prestigious aesthetic clinics in Mexico City, is expected to promote a high level of service in regenerative medicine throughout the country.
As part of this effort, the International Society for the Application of Stem Cells (ISSCA) has granted Dr. Vanessa Rodriguez Peers affiliation and use of their brand, products, therapies and training on how to apply stem cell therapies.
“This new partnership aims to expand the Global Stem Cells Group (GSCG) brand and create centers of excellence in cell therapy to meet the high demand in the Mexican market,” said Benito Novas, CEO of Global stem cells group “GSCG is rapidly expanding its global operations as it seeks to become a major player in the lucrative regenerative medicine industry. To achieve our expansion plans, our organization is partnering with healthcare providers specializing in regenerative medicine with at least five years of experience in the healthcare sector.”
Stem cell therapy is becoming an increasingly effective clinical solution for treating conditions that traditional or conventional medicine only offers within palliative care and pain management. Patients around the world are seeking a natural regenerative alternative without the potential risks and side effects sometimes associated with conventional pharmaceuticals.
The opening of this center, which will include the construction of an autologous tissue processing laboratory and an allogeneic tissue bank, will help stem cell therapy and regenerative medicine finally move from being an elective procedure to being accessible to patients throughout Mexico.
About Dr. Vanessa Rodriguez Pares
Dr. Vanessa Rodriguez Pares is a specialist in aesthetic medicine and surgery, with special attention to obesity and overweight in all ages. Management of cosmetic surgery with a comprehensive approach to the patient, taking into account their safety and their physical, psychological and emotional needs. Specialist in ULTHERAPY treatment to perform facelift without surgery. In addition to extensive experience with cellular therapies and regenerative medicine since 2016, specializing in anti-aging techniques, hair regeneration, facial aesthetics with stem cell assisted lipotransfer among other techniques.
About the Global Stem Cell Group
Global Stem Cells Group, Inc. is the parent company of six wholly owned operating companies dedicated entirely to stem cell research, training, products, and solutions. Founded in 2012, the company combines dedicated researchers, physician and patient educators, and solution providers with the shared goal of meeting the growing worldwide need for leading edge stem cell treatments and solutions. With a singular focus on this exciting new area of medical research, Global Stem Cells Group and its subsidiaries are uniquely positioned to become global leaders in cellular medicine.
Global Stem Cells Group’s corporate mission is to make the promise of stem cell medicine a reality for patients around the world. With each of GSCG’s six operating companies focused on a separate research-based mission, the result is a global network of state-of-the-art stem cell treatments.
Global Stem Cells Group is a publicly traded company operating under the symbol MSSV. https://finance.yahoo.com/quote/mssv/
To learn more about Global Stem Cells Group, Inc.’s companies visit our website
www.stemcellsgroup.com or call +1 305 560 5331
- Published in News
ISSCA Announces the Regenerative Medicine World Congress Speaker list and Promgram
The International Society for Stem Cell Application (ISSCA) has announced the Regenerative Medicine World Congress 2022 that will take place at the Radisson Blu Hotel in Istanbul, Turkey, between September 23rd and 25th, 2022. The congress brings together world-class doctors and scientists from over 35 countries.
Participants at the congress will have a rare chance to listen to over 25 world-leading medical experts as they make evidence-based research presentations on various issues related to stem cell therapy and regenerative medicine via evidence-based presentations.
Highlights of the Congress
Interactions with World-Class Speakers
Participants at the congress will have a chance to engage scientists and physicians with extensive experience applying cellular therapies and regenerative medicine in clinical environments. It will be an opportunity to gain insights into the industry and applications of this technology.
Updates on the Latest Products and Technologies in Regenerative Medicine
Some items to be discussed at the congress include new technologies, the latest products and equipment in regenerative medicine, and cellular studies. Participants will access these technologies and understand how they play a part in developing solutions to different problems in the medical field.
Hands-on Training by Leading Instructors
Participants will get two days of intensive training on clinical and aesthetic applications of stem cell therapy as part of the workshop. This training will be provided by ISSCA-certified instructors with experience in the particular areas that they will be handling. On the third day. participants will participate in live practical lessons on areas lectured on in the last two days. However, the practical lessons will be limited to 20 seats and available only to participants who purchase the all-access package.
An Opportunity to Network
Doctors and scientists will get an opportunity to network with like-minded professionals from different parts of the world to expand their knowledge and be part of a growing team of innovative minds who are leading the future of medicine. Participants will have the opportunity to interact, share ideas, and contact fellow professionals and speakers at the congress.
The Program
The president of ISSCA. Dr. Dayeong Kim (Korea). will open the program before the rest of the instructors take the stage. This team will tackle diverse areas that show the use of stem cell therapy in various medical conditions and real-world cases where the technology is being applied. Participants will get a chance to gain insights and expand their thoughts on the matters surrounding the use of stem cell technology to deal with various medical conditions.
Conference Packages
There are two packages for the conference. The first package will give participants access to two-day lectures on September 23rd and 24th. This package is going for $499.
The second package, dubbed “All-Access.” will include the one-day hands-on certification at ReGen iC Clinic on September 25th. in addition to two two-day lectures. This package will go for $950.
There are only 20 seats in the All-Access package. You can reserve a seat in either of the packages by purchasing the ticket on the ISSCA website.
More information regarding the conference can be found on the ISSCA website.
- Published in News
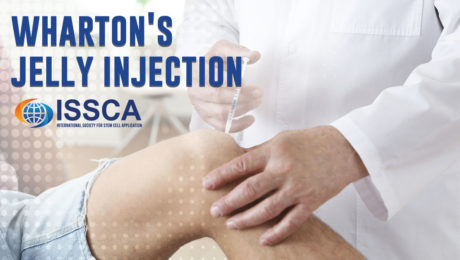
Wharton’s Jelly injection
MONDAY, 20 SEPTEMBER 2021 / PUBLISHED IN BLOG
What is Wharton’s Jelly?
Wharton’s Jelly is the gelatinous connective tissue found in the umbilical cord. This once discarded substance, previously thought of as afterbirth waste, is rich in healing substances and abundant in mesenchymal stem cells (MSCs).
Benefits of Mesenchymal Stem Cells from Wharton’s Jelly
The mesenchymal stem cells in Wharton’s Jelly are special because they are not readily recognized by the body’s immune system. They are considered “primitive cells,” meaning they have properties similar to embryonic cells. This reduces the risk of an immune reaction when these cells are injected into a patient.
Minimally processed Wharton’s Jelly preserves the growth factors and proteins needed for effective healing.
Who Can Benefit from Wharton’s Jelly Treatment?
Wharton’s Jelly is used in regenerative medicine, which employs naturally occurring cells and substances to promote tissue regeneration and healthy cell growth. This natural substance contains collagen, anti-inflammatory properties, growth proteins, and hyaluronic acid.
Treatment with Wharton’s Jelly can help with various ailments, including:
- Degenerative diseases
- Osteoarthritis
- Ligament and muscle damage
- Joint pain and inflammation
- Chronic pain
- Wound healing
There are no age restrictions on who can receive this treatment, and often a patient will only need treatment once.
How Does Wharton’s Jelly Treatment Work?
A specialized, minimally processed product derived from Wharton’s Jelly is injected into the injured site. While numbing is not always needed, lidocaine may be used for particularly sensitive areas. This process involves a relatively painless pinch of a needle. Once injected, the mesenchymal stem cells begin to transform into the cells needed to regenerate damaged tissue, reducing inflammation and pain, and restoring functionality.
Are There Any Side Effects to the Treatment?
No long-term risks or side effects have been reported. It is important to discuss proper protocols with your doctor before treatment.
Reasons to Undergo Cellular Therapy in Cancun, Mexico with Us
One of the main advantages of receiving cellular therapy treatment in Mexico is the ability to cultivate and reproduce, in a laboratory environment, the cells extracted from patients. This means a patient can receive a much larger concentration of cells, leading to improved results.
Additionally, Mexico is a great option for treatments that require a more invasive approach—a larger operating team, more medical products, and PPE. As one of the Western Hemisphere’s largest medical tourism capitals, treatments in Mexico are performed in hospitals that comply with FDA regulations and are offered at a more affordable price point.
While these treatments require a large investment from the patient, the cost of a multiple-day cellular therapy treatment in Mexico is far more affordable than the same treatment in the United States and Canada, especially without the ability to use culture-expanded cells.
What is the Recovery Period?
The actual injection does not require a recovery period, and most patients can return to their normal daily activities immediately. In cases of injury to ligaments or joints, rest may be required. Please follow your doctor’s advice for the best results from treatment.
Overall, Wharton’s Jelly injection treatment is minimally invasive and can help with a variety of ailments. With its limited side effects and regenerative properties, it is safe for patients of any age.

Contact Us
Talk with us to see if Wharton’s Jelly injection therapy is the right fit for you: Click here
About Cellular Hope Institute
At Cellular Hope Institute, we evaluate, diagnose, and treat patients around the world on a daily basis with the latest regenerative medicine modalities available today.
- Published in Blog
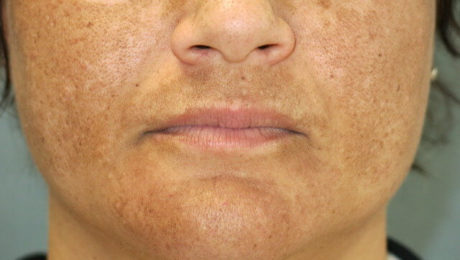
Platelet-Rich Plasma For Melasma — Will It Fade Forever?
WEDNESDAY, 18 APRIL 2018 / PUBLISHED IN BLOG
For most women, a tiny pimple on the face is enough to ruin their day or week. Even the slightest imperfection that may have a 1% chance of getting noticed by others can be distressing. For these women, melasma is their darkest nightmare. It’s a common issue, often resulting from sun exposure, that causes brown patches on the face. Unfortunately, these patches are often permanent.
The Typical Journey of Melasma Sufferers
If you’re suffering from melasma, the road to “recovery” usually looks like this:
- Hope for Natural Fading: You hope that it’ll fade away on its own.
- Home Remedies: Your friend suggests you try apple cider vinegar and lemon juice treatment.
- Dermatologist Visit: Slightly disappointed, you visit a dermatologist who’ll prescribe a bleaching cream (hydroquinone or similar).
- Full-on Disappointment: The treatment doesn’t work as expected.
- Research: You Google the hell out of the topic and feel overwhelmed by the information.
- Concealers and Makeup: Concealers and makeup become your best friends to cover the patches.
At this point, no one can convince you there is a treatment for getting rid of melasma. Trying more and more treatments only runs the risk of making the condition worse. So, what would you do?
Platelet-Rich Plasma for Melasma
What is Platelet-Rich Plasma (PRP)?
According to recent Turkish and Malaysian studies, Platelet-Rich Plasma is showing great promise for melasma. One of the good things about PRP for melasma is that it won’t make the condition worse, unlike IPL, Fraxel, or other treatments. It’s like getting a natural facial treatment with a whole lot of potential benefits, even if it doesn’t help cure melasma.
How Does PRP Work for Melasma?
PRP injections work by supplying growth factors to reduce pigmentation. Being an independent treatment with no downtime, it can be done in conjunction with conventional treatments for melasma to add and enhance effects. PRP contains more than 30 bioactive substances that play roles like increasing skin volume and adding new blood vessels.
Platelet-Rich Plasma with Microneedling
Combining PRP with Microneedling
This is the most common combination for Platelet-Rich Plasma therapy. The combination has produced results for a variety of skin pigmentation issues, making it a valuable option for melasma, especially when creams and peels haven’t helped. More importantly, PRP’s ability to stimulate collagen production in the area tightens the pores and makes your skin glow.
Why Platelet-Rich Plasma?
The Science Behind PRP
PRP is primarily a healing vehicle. It needs to be injected into the membrane below the skin. It works by supplying the underlying skin membrane with collagen and tenascin stimulated by the transforming growth factors in PRP. These growth factors also promote the formation of new blood vessels, sometimes resulting in the disappearance of spider veins.
The released growth factors (mainly platelet-derived growth factor (PDGF), epidermal growth factor (EGF), vascular endothelial growth factor (VEGF), fibroblast growth factor (FGF), and transforming growth factor-beta (TGF-ß)) stimulate the proliferation of fibroblasts and epidermal cells, and collagen synthesis. Additionally, TGF-ß has been proven to inhibit melanogenesis — or reverse skin pigmentation — the exact opposite effect of exposure to UV-B radiation.
Treatment Results and Maintenance
Typically, patients see excellent results with 2-3 PRP injections in the first 3 months. Clinical studies have shown that the effects maintain after 6 months. However, melasma is known to recur even after successful treatments. Therefore, you must take precautions by using sunscreen with broad-spectrum protection and an SPF of 30 or higher. Avoiding harsh skincare products is also essential as they can exacerbate melasma.
By structuring the article with H2 headings and optimizing readability, this version should be more engaging for readers and better optimized for search engines.
- Published in Blog