Stem Cell Research and Stem Cell Therapy: When can stem cells be used to treat patients?
The difference between stem cell research and therapy is in the scientific evidence that supports therapeutic intervention to be beneficial for the patient.
Stem cells have the remarkable potential to develop into many different types of cells in the body during early life and growth. In addition, in many tissues, stem cells serve as a sort of internal repair system, dividing essentially without limit to replenish other cells as long as the individual is alive. When a stem cell divides, each new cell has the potential either to remain a stem cell or become another type of cell with a more specialized function, such as a muscle cell, a red blood cell, or a brain cell.
Stem cell research on adult stem cells
Stem cells are distinguished from other cell types by two important characteristics. First, they are unspecialized cells capable of renewing themselves through cell division, sometimes after long periods of inactivity. Second, under certain physiologic or experimental conditions, they can be induced to become tissue- or organ-specific cells with special functions. In some organs, such as the gut and bone marrow, stem cells regularly divide to repair and replace worn out or damaged tissues. In other organs, such as the pancreas and the heart, stem cells only divide under special conditions.
Until recently, scientists primarily worked with two kinds of stem cells from animals and humans: embryonic stem cells and non-embryonic “somatic” or “adult” stem cells in stem cell research.
In 2006, researchers made a breakthrough by identifying conditions that would allow some specialized adult cells to be “reprogrammed” genetically to assume a stem cell-like state. This new type of stem cell is called induced pluripotent stem cells (iPSCs).
Stem cells are important for living organisms for many reasons. In the 3- to 5-day-old embryo, called a blastocyst, the inner cells give rise to the entire body of the organism, including all of the many specialized cell types and organs such as the heart, lungs, skin, sperm, eggs and other tissues. In some adult tissues, such as bone marrow, muscle, and brain, discrete populations of adult stem cells generate replacements for cells that are lost through normal wear and tear, injury, or disease.
Stem cell research for treating disease
Given their unique regenerative abilities, stem cells offer new potentials for treating diseases such as diabetes, and heart disease. However, much work remains to be done in the laboratory and the clinic to understand how to use these cells for cell-based therapies to treat disease, which is also referred to as regenerative or reparative medicine.
Laboratory studies of stem cells enable scientists to learn about the cells’ essential properties and what makes them different from specialized cell types. Scientists are already using stem cells in the laboratory to screen new drugs and to develop model systems to study normal growth and identify the causes of birth defects.
Research on stem cells continues to advance knowledge about how an organism develops from a single cell and how healthy cells replace damaged cells in adult organisms. Stem cell research is one of the most fascinating areas of contemporary biology, but, as with many expanding fields of scientific inquiry, research on stem cells raises scientific questions as rapidly as it generates new discoveries.
In 1964, the World Medical Association developed the Declaration of Helsinki as a statement of
ethical principles for medical research involving human subjects. It includes research on identifiable human material and data, last amended in October 2013.
According to the Helsinki Declaration, in the treatment of an individual patient where proven interventions do not exist or other known interventions have been ineffective, the physician, after seeking expert advice, with informed consent from the patient or a legally authorized representative, may use an unproven intervention if in the physician’s judgement it offers hope of saving life, re-establishing health or alleviating suffering.
Intervention should subsequently be made the object of research, designed to evaluate its safety and efficacy. In all cases, new information must be recorded and, where appropriate, made publicly available.
- Published in Blog
Could stem cells offer the cure for muscular dystrophy?
The term muscular dystrophy (MD) refers to a group of disorders in which a genetic abnormality causes muscles responsible for controlling movement to become weak, and muscle mass to be lost. These inherited disorders usually affect voluntary (skeletal) muscles, although weakness can also extend to the muscles that control respiration and swallowing.
Given that the genetic mutations triggering MD interfere with the normal production of certain critical proteins, the body is not able to reverse muscle weakening or loss of mass, so even when the disease progresses slowly, it eventually affects one’s ability to walk in a more or less conducive manner.
Who is affected by muscular dystrophy?
In most cases MD appears in infancy, but it’s not uncommon for symptoms to start manifesting in teens or adults.
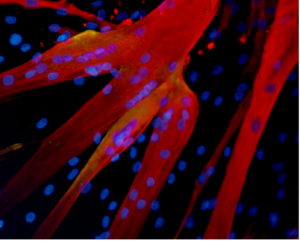
Muscle fibers formed in the lab by human mesoangioblasts (image: eurostemcell).
There are different kinds of muscular dystrophy, the most common and severe form being Duchenne muscular dystrophy (DMD) Caused by a genetic flaw or defect, Duchenne MD is more common in males than females [1} and affects about 1 in every 3,500 boys worldwide.
The onset of Duchenne muscular dystrophy occurs between the ages of 2 and 6, and evolves slowly. Muscles becoming weaker year after year, and the spine and limbs becoming progressively deformed. In most cases, children affected by this form of the disease become wheelchair dependent by the age of 12.
People suffering from Duchenne MD often die in their 20s, and those who survive usually experience some degree of cognitive impairment. The shortening of tendons and muscles limits the mobility of sufferers even more, and breathing and heart problems can occur.
Treatments for Duchenne muscular dystrophy
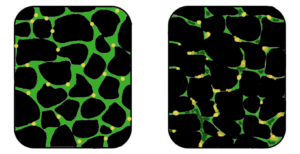
Muscular dystrophy is a genetic disorder where the muscle tissue wastes away and loses function. In the affected muscle (left), the tissue has become disorganized and the concentration of dystrophin (green), an important protein in normal muscle functioning, is greatly reduced. (Image: Wikipedia)
In a healthy organism, damaged muscles repair themselves thanks to a series of cells that include muscle stem cells, called satellite cells. In Duchene muscular dystrophy, the muscles lack dystrophin, the protein needed for maintaining the integrity of muscle fibers. Without this protein, the burden placed on the body’s naturally occurring muscle stem cells is too intense, rendering the cells unable to repair damaged muscle tissue or to generate new muscle mass to replace wasted mass [6].
For this reason, scar tissue and fat cells take the place of damaged muscle tissue, contributing to muscle weakening and, over time, cause muscles to lose their functional ability. Would it be possible for the damaged muscle fibers to regain their regenerative ability with help from transplanted stem cells?
Research suggests stem cells could be a potential solution for muscle wasting
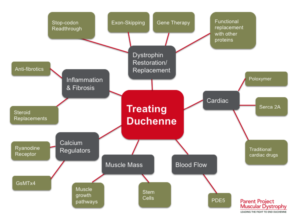
(Click on image to enlarge) Considerable efforts are underway to develop drugs and biologics (cell and gene therapy) to address the primary problem in Duchenne—the absence of dystrophin. Restoring dystrophin or replacing dystrophin with replacement protein are considered foundational therapies.
In 2006, researchers managed to restore mobility in two afflicted dogs using stem cells isolated from muscle blood vessels [4], and in 2007 scientists managed to treat Duchenne MD in research mice using a combination of genetic correction and stem cells [3]. The latter study showed that it is possible to correct the genetic error in the cells that no longer produce dystrophin protein, and inject corrected cells stimulating the regeneration of muscles.
Researchers at the Harvard Stem Cell Institute obtained similar results, demonstrating that transplanted muscle stem cells can improve function in mice with MD, while replenishing the stem cell population in muscle fibers [5].
Although it’s still too early to say whether stem cells can cure DMD in humans, it’s clear that there are some promising stem-cell-based approaches for Duchenne MD. One solution is to replace the defective stem cells with healthy stem cells, as these may be able to generate working muscle fibers to replace damaged muscle fibers .
A second solution would be to reduce the inflammation that speeds up the loss and weakening of muscles using certain types of stem cells [2]. Combined treatments, such as mixing stem cell therapies with gene therapies are also being tested and may prove successful in the near future.
References:
- http://www.mayoclinic.org/diseases-conditions/muscular-dystrophy/basics/definition/con-20021240
- http://www.eurostemcell.org/factsheet/muscular-dystrophy-how-could-stem-cells-help
- https://www.mda.org/disease/duchenne-muscular-dystrophy/research
- http://quest.mda.org/article/scientists-bullish-stem-cells-muscle-repair
- http://hsci.harvard.edu/stem-cells-used-treat-muscular-dystrophy-mice
- https://med.stanford.edu/news/all-news/2014/12/stem-cells-faulty-in-duchenne-muscular-dystrophy.html
http://www.mayoclinic.org/diseases-conditions/muscular-dystrophy/basics/definition/con-20021240
- http://www.eurostemcell.org/factsheet/muscular-dystrophy-how-could-stem-cells-help
- https://www.mda.org/disease/duchenne-muscular-dystrophy/research
- http://quest.mda.org/article/scientists-bullish-stem-cells-muscle-repair
- http://hsci.harvard.edu/stem-cells-used-treat-muscular-dystrophy-mice
- https://med.stanford.edu/news/all-news/2014/12/stem-cells-faulty-in-duchenne-muscular-dystrophy.html
- Published in Blog
- 1
- 2

